Mosquito Repellent
Mosquito repellents are substances that are designed to make surfaces unpleasant or unattractive to mosquitos. They typically contain an active ingredient that repels mosquitos as well as secondary ingredients, which aid in delivery and cosmetic appeal. They are available in many forms, from creams to lotions to oils, but are most often sold as aerosol products.
History
Traditionally, various types of substances have been used to repel mosquitos. These include such things as smoke, plant extracts, oils, tars, and muds. As insect repellent technology became more sophisticated, individual compounds were discovered and isolated. This allowed the formulation of new, more efficient forms of mosquito repellents.
The first truly effective active ingredient used in mosquito repellents was citronella oil. This material is an herbal extract derived from the citronella plant, an Asian grass. While citronella had been used for centuries for medicinal purposes, its repellence was only accidentally discovered in 1901, when it was used as a hairdressing fragrance. Since citronella oil is a fragrant material, it is thought that the chemical terpenes of which it is composed are responsible for its repellent activity. Citronella oil does repel mosquitos, but it has certain characteristics which limit its effectiveness. For example, it is very volatile and evaporates too quickly from surfaces to which it is applied. Also, large amounts are needed to be effective.
The disadvantages of using citronella oil prompted researchers to study alternative synthetic compounds. Many of the early attempts at creating synthetic insect repellents were initiated by the United States military. Out of this research came the discovery of the repellent dimethyl phthalate in 1929. This material showed a good level of effectiveness against certain insect species, but it was ineffective against others. Two other materials were developed as insect repellents. Indalone was found to repel insects in 1937, and Rutgers 612 (2-ethyl-1,3-hexane diol) was synthesized soon after. Like dimethyl phthalate, these materials had certain limitations which prevented their widespread use.
Since none of the available materials were ideal repellents, research into new synthetic materials continued. In 1955, scientists synthesized DEET (n-n-diethylnetatoluamide), currently the most widely used active ingredient for mosquito repellents. After its discovery, repellent manufacturers developed many different forms in which to deliver DEET, such as creams, lotions, and aerosols.
Mode of Action
Most repellent chemicals work by interfering with the mosquito's homing system. This homing system, located on the antennae, is made up of a number of chemical receptors. Research has shown that these chemical receptors are activated by lactic acid, which naturally evaporates from the skin of warm-blooded animals. The mosquitos have the innate ability to follow the lactic acid emissions to their source. However, when a repellent ingredient such as DEET is applied to the skin, it also evaporates. It is thought that the chemical inhibits the binding of the lactic acid to the mosquito's chemical receptors. This essentially "hides" the protected person from the mosquito. Since the active ingredient must evaporate from the surface to work, the repellent activity lasts for a limited time.
Raw Materials
The active ingredient in a mosquito repellent is primarily responsible for its usefulness. For a material to be valuable as a mosquito repellent, it must meet certain criteria. First, it must effectively discourage insect attack on the treated area for many hours and on many different types of surfaces. Second, it must work under a variety of different environmental conditions. Next, it must not be toxic or cause irritation when applied to human or animal skin. Additionally, it must be cosmetically acceptable, having a pleasant odor, taste, and feel. It should also be harnless to clothing. Finally, it should have a relatively low cost and be effective against other common types of insects, such as flies.
While thousands of compounds have been studied for their use as insect repellents, DEET (n, n-diethyl-m-toluamide) has been used more than any other. DEET is the compound which results from a reaction of m- toluic acid with thionyl chloride followed by a reaction with diethyl amine. This material is isolated and purified before it is supplied to mosquito repellent manufacturers. Other repellent ingredients used include citronella oil, dimethyl phthalate, lavender, lemon-grass oil, and peppermint oil. It has been found that mixtures of various repellent compounds often provide greater effectiveness than any one compound alone. The active ingredients contained in the mosquito repellents generally make up 5-30% of the final products.
The inert ingredients that are in a mosquito repellent depend on the form that the product will take. Currently, mosquito repellents are sold as aerosols, pumps, lotions, and oils. Mosquito repellents that are sold as lotions or creams are essentially skin creams which have DEET added at a certain level. They are primarily composed of water, surfactants, fatty alcohol, fragrance, and other emollients. When applied to the skin, these products have the dual benefit of repelling mosquitos and moisturizing skin. These products are generally less effective than aerosol forms, however, because they do not allow the active ingredients to evaporate as easily.
Aerosols are the most common form for mosquito repellents. They are made up of a few different types of ingredients, including a solvent, a propellant, and miscellaneous ingredients. The solvent is usually an organic alcohol such as ethanol or propanol, whose primary responsibility is to dilute the active ingredient to an appropriate concentration. It also aids in keeping all of the raw materials mixed, ensuring that the product will remain effective even after long-term storage. The propellant is a volatile compound which creates the pressure that causes the rest of the product to be forced out of the container. Common propellants include liquified hydrocarbon gases like propane, butane, or isobutane, hydrofluorocarbons, and dimethyl ether. Other ingredients such as fragrances and emollients are added to aerosol mosquito repellents to make them more cosmetically appealing. Still other compounds are added to prevent corrosion and other stability problems.
In addition to the ingredients, the packaging components are also an important part of an aerosol mosquito repellent. The can is typically a metal container made up of tin-plate steel. The coating of tin keeps the steel from reacting with the ingredients used in the repellent formulation. The valve is another key packaging component. It has the dual task of sealing the pressurized contents in the can and controlling the dispensing of these contents. Valves have three sections: a diptube, which feeds the product from the can to the valve body; the valve body, which mixes the product and propellant; and the actuator button, which when pressed, allows the product to be released.
The Manufacturing
Process
The production of mosquito repellents can be broken down into two steps. First a large batch of the repellent formulation is made, and then the batch is filled into the packaging. Since aerosols are the most common form of mosquito repellent, the following

Compounding
- 1 The first step in the manufacturing process is compounding. In the compounding area, raw materials are mixed together in large stainless steel tanks. For an aerosol, the alcohol is pumped into the tank, and the other materials, including DEET, fragrance, and emollients, are manually poured in and allowed to mix. All of the ingredients except the propellant are added at this phase of production. Since some of the materials in this process are flammable, special precautions are taken to prevent explosion, such as using spark-proof electrical outlets and blast-proof walls.
- 2 When the batch is finished, a sample is sent to the quality control lab and tested to make sure it meets the set standards for the product. After passing these tests it is pumped to the filling lines to make the finished product.
Filling
-
3 The filling line is a series of machines connected by a conveyor belt
system that combine all of the components to make the finished mosquito
repellent product. The first machine in the system feeds the empty cans
onto the conveyor line. This machine has a large hopper that is filled
with empty
cans which are physically manipulated until they are standing upright and correctly oriented.
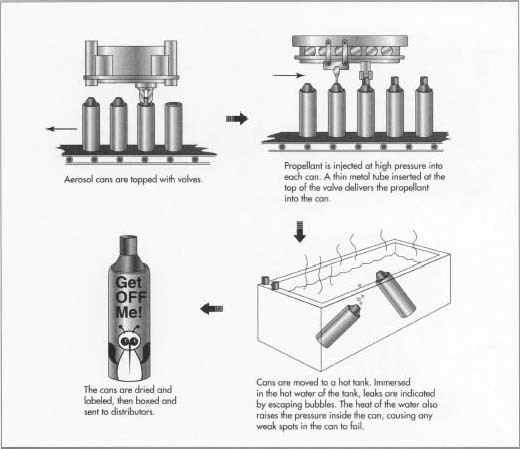
- 4 The metal cans are then automatically moved along the conveyor belt and cleaned with a jet of compressed air to remove any dust and debris. They next travel to the filling carousel. The filling carousel is made up of a series of piston filling heads that are calibrated to deliver exactly the correct amount of product into the cans. As the cans move through this section of the filling line, they are filled with product.
- 5 The next step in the filling process involves topping the cans with a valve, adding the propellant, and pressurizing the cans. The valve is put on by the valve inserter machine. Much like the bin that holds the empty cans, the valves are also put in a hopper and then correctly sorted and aligned. As the cans pass by, the valves are put on. These valves are then tightly affixed to the can by the valve crimping machine. Depending on the type of filling technique, the propellant is either injected through the valve at high pressure or injected into the can before the valve is crimped.
- 6 After the cans are capped and filled, they are moved to a hot tank, a long trough filled with hot water. Here the cans are checked for escaping bubbles that would indicate a propellant leak. The high temperature of the waterbath also raises the internal pressure of the can, which is intended to cause any weak spots in the can to fail. This is a crucial quality control step that prevents damaged cans from being sold to the public.
- 7 When the cans exit the waterbath, they are dried by high-pressure air jets. Other components are then added, such as the actuating button and the overcap. Any needed labels or printing are also added at this point.
- 8 The finished cans are then moved to the boxing area, where they are put into boxes, typically a dozen cans at a time. These boxes are then stacked onto pallets and hauled away in large trucks to distributors. High speed aerosol production lines like the one described can move at speeds of about 200 cans per minute or more.
Quality Control
Quality control is an essential step in the production of mosquito repellents. Tests are performed at various points in the manufacturing process to ensure that the finished products are consistent from run to run, remain effective over a long period of time, and are safe to use.
Before production begins, the incoming raw materials are checked to ensure they meet the previously set specifications. Tests such as pH, specific gravity, and moisture content are typically performed. Additionally, the cans are inspected for dents, corrosion, and other weaknesses. During manufacture, samples of the repellent are taken during different points along the filling line, and the characteristics or the product are tested. Some of the monitored parameters include the level of active ingredient, pressure, spray rate, and spray pattern. Other testing is conducted to ensure that the cans evacuate properly. Also, long-term stability studies may be done to establish that the cans do not show undue signs of corrosion.
The Future
The use of many available mosquito repellents is not without its drawbacks. Products that use DEET or citronella oil as the primary active ingredients have been reported to causes rashes in some people. There have even been cases in which children who used DEET products have become very ill. For this reason, research has focused on finding new types of repellents and methods for improving the safety of the ones that are currently available. One recent advance in repellent technology is the use of chemicals to "encapsulate" DEET. It is thought that this product form will protect the user from the harmful effects of DEET while still maintaining its repellent activity. More investigation will have to be completed before this is verified.
Where to Learn More
Books
Knowlton, J. and S. Pearce. Handbook of Cosmetic Science and Technology. Elseveir Science Publications, 1993.
Periodicals
Holmes, Hannah. "The Battle of the Bug." Backpacker, April 1996, pp. 68-72.
Romanowski, P. and R. Schueller. "Aerosols for Apprentices." Cosmetics & Toiletries, May 1996, pp. 35-40.
— Perry Romanowski
Comment about this article, ask questions, or add new information about this topic: