Rubber Cement
Background
Rubber cement is a solution of unvulcanized (gum) rubber in a solvent, and is used as an adhesive. Ideally, it is meant to join two pieces of rubber together, which involves a chemical cohesion process. When joining two pieces of rubber, only one surface has to be coated with rubber cement since they are the same material. However, when joining paper together, both pieces need to be covered with rubber cement. When rubber cement dries, only the parts in contact with the paper remain, which holds the two pieces together. Despite this limitation, the household type of rubber cement finds wide use for applications such as mounting photographs. Unlike with white glue, the joined pieces of paper can be pulled apart without damaging either piece.
Adhesives are made from either natural animal or plant products or synthetic polymer. Natural adhesives are easy to apply and in general are water soluble. Synthetic adhesives are divided into four chemical categories: thermoplastic, thermosetting, elastomeric, and combinations thereof. Thermoplastic adhesives, such as polyvinyl alcohol and acrylics, can be resoftened since the materials do not crosslink upon curing. They require heat or a solvent to create a bond. Thermosetting adhesives, which include epoxies, cannot be heated and resoftened after curing because they do crosslink upon curing. Elastomeric adhesives are based on isoprene rubber or synthetic polymers that combine both elasticity and toughness. Silicone is a typical example.
The properties of the adhesive, the types of materials to be joined, and the condition of the surfaces all determine the performance of the joints and the service life of the bonded structure. The adhesives must be able to wet and spread properly on either surface to achieve molecular contact between the materials. Adhesives are used in a wide range of industries, including packaging, construction, electronics, transportation, furniture/woodworking, and medical.
In 1996, over 12 billion lb (5.4 billion kg) of adhesives were used in the United States. Construction applications had the highest share at 40%. This is also one of the largest applications for rubber cement. Other applications include heating, air conditioning, and automotive equipment. Manufacturers of rubber cement also sell a lot of their product to repackagers who market the product under their own name.
Some analysts put the global market for adhesives at $19.1 billion in 1997. Packaging, construction, and furniture/woodworking are the three largest segments of this global market, with over 65% of market revenues.
History
Natural adhesives have been around for at least several thousand years. Egyptian carvings show the gluing together of thin pieces of veneer to a wooden plank. Fibers in ancient fabric were joined together with flour paste, and gold leaf was bonded to paper with egg white. Animal glues improved during the eighteenth century and a century later, rubber-and nitrocellullose-based cements were introduced. During the 1900s, significant advances occurred, leading to the development of many synthetic adhesives that replaced some of the natural adhesives. Adhesives now had to be much stronger and more corrosion resistant.
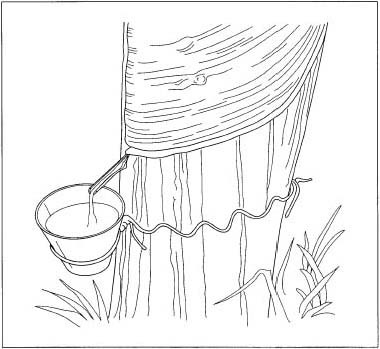
Natural rubber was first discovered by the Mayas and Aztecs over 2,500 years ago and used to make shoes and clothing waterproof. Centuries later in 1823, the Scottish chemist Charles Macintosh also investigated making waterproof textiles. He experimented with dissolving rubber in various chemicals and had the most success with naphtha. With this material he made a rubber paste and used it to join together two layers of cloth to make a raincoat.
Raw Materials
Rubber cement is an opaque liquid that contains pulverized natural or synthetic rubber and a solvent based on hexane or heptane. Grades of rubber cement may contain 70-90% heptane or hexane and 1-15% isopropyl alcohol (isopropanol) or ethyl alcohol (ethanol). The rubber is received in the form of large blocks or slabs, typically 100 lb (45 kg) in size. Thousands of gallons (liters) of liquid solvents are usually shipped by tank truck to the manufacturer.
Natural rubber comes from the Hevea brasiliensis tree originally found in Brazil. To make solid rubber, the tree is tapped and the latex is collected in a small cup, where it coagulates into a lump. This lump, together with the leftover flow and other pieces are collected together and processed at high temperature. This destroys most of the proteins and produces a solid material.
Synthetic rubbers include neoprene and latex. Synthetic rubbers are made using various chemical processes. The application determines what types of rubber and solvent are used.
Design
The properties and performance of the rubber cement are determined by the type and amount of ingredients. Typically, formulations are determined by the laboratory and then given to production.
The Manufacturing Process
The process to make rubber cement is relatively simple. After the rubber is broken down into smaller pieces, it is mixed with the hexane-or heptane-based solvent and then various sizes of containers are filled with the liquid. Most equipment is automated.
Mixing
- 1 First, the blocks or slabs of rubber are broken into smaller pieces. Rubber mills, equipped with two large rollers, are used. The rubber may be reduced in large high speed mixers equipped with sharp blades, which pulverize the rubber into a size similar to sawdust. The solvent ingredients are mixed in tanks, ranging from 40 to 6,000 gal (150 to 22,700 1), equipped with paddles. The rubber is added slowly until it is wetted by the solvent and is suspended or dissolved in the solution.
Packaging
- 2 Once the solution is thoroughly mixed it is fed into an automated filling line, which fills each container, caps it, and tightens the cap. Rubber cement is packaged in 4, 8, and 16 oz (118, 237, and 473 ml) bottles or quart (946 ml) and gallon (3.8 1) containers. Because the rubber cement is flammable, explosion proof equipment is used. The containers are then packaged in cardboard cases and properly labeled. For larger containers (tank trucks or drums), the solution is gravity fed or pumped into these containers from the bottom.
Quality Control
The raw materials are supplied according to the manufacturer's specifications. Each batch is checked for solids content, since better quality grades contain higher content. The percent solids content is obtained by weighing a sample, evaporating the liquid, and then weighing the remaining solid. After mixing, each batch is also tested for viscosity, tackiness, heat load, and other properties before being packaged. Up to 20 different tests may be performed. After packaging, the containers are checked to make sure they are properly labeled.
Byproducts/Waste
Because of the tight controls and automated equipment, no waste is produced. The filling machine is programmed for the exact volume of each size of container. Any leftover material is recycled back into the process. Rubber cement is shipped with a material safety data sheet (MSDS) that outlines proper handling procedures since the ingredients are hazardous and flammable. The solvent is a volatile organic compound and is also subject to regulation as a hazardous air pollutant under the 1990 Clean Air Act Amendments. The U.S. Environmental Protection Agency also has included adhesives as one of the target categories that must comply with certain Maximum Achievable Technologies (MACT). A MACT sets a level of control designed to protect public health.
The Future
Demand for all adhesives in the United States is expected to reach over 15 billion lb (6.8 billion kg) by 2003, a 2.6% annual growth rate. The global market is expected to reach $26.2 billion in 2003, with a compound annual growth rate of 5.3%. Solvent-based adhesives will continue to be replaced by water-based adhesives since they are more environmentally friendly.
Where to Learn More
Other
Starkey Chemical Process Co. P.O. Box 10, 9600 W. Ogden Ave., LaGrange, IL 60525-2534. (708) 352-2565.
— Laurel M. Sheppard
is it possible to have the price of a natural rubber manufacturing company ? for a monthly average production of 200 tons ?
Thank you Sirs.
I want to know about how can I adhere two pieces of elastik rubber band.
rubber bands have 40 shoreA hardness and 700% elongation at break.
can you please help me?