Latex
Background
A latex is a colloidal suspension of very small polymer particles in water and is used to make rubber.
Natural
Dipped goods (medical and surgical items, household and industrial gloves, boots, and balloons) utilize more than half of all natural latex consumed in the United States. The adhesives industry is the second largest user of natural latex in products such as shoes, envelopes, labels, and pressure sensitive tape.
Natural latex with a high solids content is also used for making molds for casting plaster, cement, wax, low temperature metals, and limited run polyester articles. Natural latex has the ability to shrink around the object to be reproduced, so that the smallest detail will be reproduced in the cast. Latex is even being used to help stabilize desert soils to make them suitable for agricultural uses.
Natural latex is produced from the Hevea brasilienesis rubber tree and is the protective fluid contained beneath the bark. It is a cloudy white liquid, similar in appearance to cow milk. It is collected by cutting a thin strip of bark from the tree and allowing the latex to exude into a collecting vessel over a period of hours.
Hevea trees mature at five to seven years of age and can be tapped for up to 30 years. Rubber yields range around a ton per acre (2.5 tons per ha) on the larger plantations, but yields four times as much are theoretically possible. Trees often are rested for a period after heavy tapping.
Natural latex was once commercially produced in the Amazon in great quantities. In recent times, production of natural latex has moved to Malaysia, Indonesia, and other Far Eastern regions. More than 90% of the total world production of natural rubber now comes from Asia, with well over half of that total originating in these countries. Other leading Asian producers include Thailand, India, and Sri Lanka. China and the Philippines both have substantially increased their rubber production as well.
Synthetic
Most synthetic rubber is created from two materials, styrene and butadiene. Both are currently obtained from petroleum. Over a billion pounds (454,000,000 kg)of this type of rubber was manufactured in the United States in 1992. Other synthetic rubbers are made from specialty materials for chemical and temperature resistant applications.
Tires account for 60-70% of all natural and synthetic rubber used. Other products containing rubber include footwear, industrial conveyor belts, car fan belts, hoses, flooring, and cables. Products such as gloves or contraceptives are made directly from rubber latex. Latex paints are essentially a solution of colored pigment and rubber latex. Latex foam is made by beating air into the latex before coagulating it.
History
The Indians of Central and South America used rubber as early as the eleventh century to coat fabric or to make into balls, but it was not until the French scientist Charles de la Condamine visited South America during
During the mid-nineteenth century, Charles Goodyear discovered vulcanization, a process that retains the rubber's elasticity under temperature changes. This process heats rubber with sulfur, which causes cross linking, decreasing rubber's tackiness and sensitivity to heat and cold.
In 1882, John Boyd Dunlop of Ireland was granted a patent for his pneumatic tire. As the demand for tires began to deplete natural rubber supplies, the British cultivated huge rubber plantations in Singapore, Malaysia, and Ceylon (Sri Lanka). Seeds were taken from Brazil and first germinated in England and then shipped to these countries. Today, all natural rubber produced in Asia comes from trees that are descendants of the Brazilian seeds.
By the early 1900s, various countries sought ways to improve rubber compounds and to develop synthetic materials. In 1910, sodium was found to catalyze polymerization. When the Germans were cut off from natural rubber supplies during World War I, they used this discovery to make about 2,500 tons (2,540 metric tons) of rubber made from dimethylbutadiene.
During World War II, the Japanese gained control of the major sources of natural rubber in Asia. In response, the United States' synthetic rubber industry increased its production by an astonishing 10,000%, from 7,967 tons (8,130 metric tons) in 1941 to more than 984,000 tons (1 million metric tons) in 1944. Following the war, other countries developed their own synthetic rubber factories to avoid having to rely on overseas rubber supplies.
Improvements in synthetic rubber have continued, and in addition, higher yielding hybrid trees have been developed that yield twice as much natural latex as the conventional ones. In 1971, a tree stimulant was developed that resulted in an average increase of 30% in latex production with no apparent harm to the trees.
Raw Materials
The composition of latex sap consists of 30-40% rubber particles, 55-65% water, and small amounts of protein, sterol glycosides, resins, ash, and sugars. Rubber has high elasticity and a polymer molecular structure. This structure consists of a long chain made up of tens of thousands of smaller units, called monomers, strung together. Each monomer unit has a molecular size comparable with that of a simple substance such as sugar. Other special chemicals are used as preservatives or stimulants during the harvesting process.
Both synthetic and natural rubber production require the use of vulcanizing chemicals, primarily sulfur. Fillers such as carbon black are also added to provide extra strength and stiffness. Oil is often used to help processing and reduce cost.
The Manufacturing
Process
Growing and processing natural rubber is one of the most complex agricultural industries and requires several years. It combines botany, chemistry, and sophisticated machinery with dexterous skills of the people who harvest the trees. Contrast this with synthetic rubber production, which involves chemical reactions and sophisticated chemical processing machinery that is automatically controlled by computers. The production of natural latex is described below.
Planting
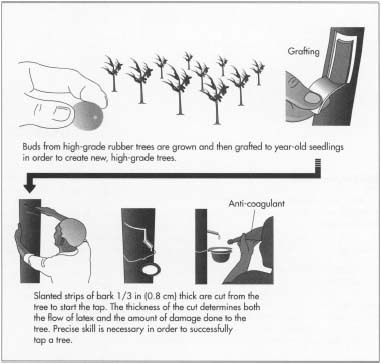
- 1 Seeds from high-grade trees are planted and allowed to grow for about 12 to 18 months in the nursery before a new bud is grafted to the seedling. After bud grafting, the year-old seedling tree is cut back and is ready for transplanting. The bud sprouts shortly after transplanting, resulting in a new tree with better properties. Approximately 150 trees are planted per acre (375 per ha), which are cultivated and cared for until they are ready for tapping in about six to seven years.
Tapping
- 2 To harvest latex, a worker shaves off a slanted strip of bark halfway around the tree and about one third in (0.84 cm) deep. Precise skill is required for if the tree is cut too deeply, the tree will be irreparably damaged. If the cut is too shallow, the maximum amount of latex will not flow. The latex then bleeds out of the severed vessels, flows down along the cut until it reaches a spout, and finally drops into a collection cup that will later be drained.
- 3 Tapping is repeated every other day by making thin shavings just below the previous cut. When the last scar created by the cuts is about 1 ft (0.3 m) above the ground, the other side of the tree is tapped in similar fashion, while the first side renews itself. Each tapping takes about three hours and produces less than a cup of latex.
- 4 A tapper first collects the cut lump, which is coagulated latex in the cup, and tree lace, which is latex coagulated along the old cut. Next, the tapper makes a new cut. The latex first flows rapidly, then declines to a steady rate for a few hours, after which it slows again. By the next day, the flow has nearly stopped as the severed vessel becomes plugged by coagulated latex.
-
5 To prevent most of the liquid latex from coagulating before it can be conveniently pooled and transported, the tapper adds a preservative such as ammonia or formaldehyde to the collection cup. Both the liquid and coagulated latex is sent to factories for processing.

- 6 To increase tree yields and reduce tapping times, chemical stimulants are used. Puncture tapping, in which the bark is quickly pierced with sharp needles, is another method that can improve productivity, since it enables the same worker to tap more trees per day.
Producing liquid concentrate
- 7 About 10% of the latex is processed into a liquid concentrate by removing some of the water and increasing the rubber content to 60%. This is achieved either by spinning the water out of the latex through centrifugal force, by evaporation, or by a method known as creaming. In this method, a chemical agent is added to the latex that causes the rubber particles to swell and rise to the liquid's surface. The concentrate is shipped in liquid form to factories, where it is used for coatings, adhesives, and other applications.
Producing dry stock
- 8 Other rubber and field latex is coagulated with acid. A giant extrusion dryer that can produce up to 4,000 lbs (1,816 kg) per hour removes the water, creating a crumb-like material. The dried rubber is then compacted into bales and crated for shipment.
Forming sheets
- 9 Ribbed smoked sheets are made by first diluting the latex and adding acid. The acid makes rubber particles bunch together above the watery serum in which they are suspended. After several hours, roughly one pound (0.45 kg) of soft, gelatinous rubber coagulates for every three pounds (1.35 kg) of latex.
- 10 The rubber is allowed to stand for one to 18 hours, then the slabs are pressed into thin sheets through a system of rollers that wrings out excess liquid. The final set of rollers leaves a ribbed pattern on the sheets that increases the surface area and hastens drying. The sheets are dried for up to a week in smoke houses before being packed and shipped.
Producing other products
- 11 To make rubber products, the mix is shaped by placing it in a heated mold, which helps shape and vulcanize the material. For more complex products, such as tires, a number of components are made, some with fiber or steel-cord reinforcement, which are then joined together. Surgical gloves are made by dipping a ceramic form into latex, withdrawing the form, and then drying the latex shape.
Quality Control
A number of quality checks are made after the latex is harvested. After tapping, the latex is checked for purity and other properties. After each step of the production process, technicians check physical properties and chemical composition, using a variety of analytical equipment.
The Future
The production of natural rubber has failed to meet the growing demand for rubber, and hence, today two-thirds of the world's rubber is synthetic. However, developments, such as the invention of epoxidized natural rubber which is produced by chemically treating natural rubber, may reverse this trend. The synthetic rubber industry is also continuing to make processes more efficient, less costly, and less polluting, as well as developing new additives, compounds, and applications.
Though there are as many as 2,500 other plants that produce rubber, it is not made fast enough to be profitable. United States Department of Agriculture researchers are looking at ways to speed up the process by genetically engineering a plant to make larger initiator molecules. These molecules start the rubber-making process, and if such molecules were larger, rubber could be produced up to six times faster.
Where to Learn More
Periodicals
Amato, Ivan. "History of Rubber Production and Use." News Service of the American Chemical Society.
"New Ways to Make Rubber Tried." The Associated Press News Service, March 24, 1995.
Other
Firestone Synthetic Rubber & Latex Company, PO Box 26611, Akron, OH 44139-0008. http://www.firesyn.com .
CEMENTEX Latex Corp., 121 Varick St., New York NY 10013. (212) 741-1770. http://www.cementex.com .
— Laurel M. Sheppard
Comment about this article, ask questions, or add new information about this topic: