Mouthwash
Mouthwash is a liquid oral product designed to freshen breath. Certain varieties may also kill bacteria and/or whiten teeth. Mouthwashes are made by combining the appropriate raw materials in large, stainless steel tanks and then filling the product into individual packages. First used by ancient societies, technological advances in chemistry have resulted in steadily improving formulas. In 1998, Americans spent over $652 million on mouthwash
Background
The need for mouthwash is a result of a condition called halitosis, or bad breath. It is estimated that over half the population occasionally has foul-smelling breath. This typically occurs upon first awakening or after a meal with garlic or onions. It has been found that bad breath is mostly due to bacterial activity in an unclean mouth. Specifically, anaerobic bacteria that grow on the protein-rich food debris stuck between the teeth or on the tongue. As the bacteria breaks down the proteins, those containing sulphur give off foul odor molecules such as methylmercaptan and hydrogen sulphide which result in bad breath.
Mouthwashes are designed to eliminate bad breath in two ways. First, they relieve it by killing the bacteria responsible for producing the foul odor. The best of these products prevent bad breath for as long as eight hours. The second way that mouthwashes help reduce bad breath is by masking the odor. This is a much less effective method which lasts no more than 30 minutes.
History
Products used for freshening breath or cleaning teeth have been in existence for centuries. Many of the ancient societies—including the Egyptians, Chinese, Greeks, and Romans—had recipes for such preparations. They used a variety of ingredients; from edible materials like fruit, honey, or dried flowers to less appealing compounds such as ground lizard, minced mice, or urine. These products were generally ineffective and in some cases were harmful to the sensitive enamel which coats each tooth.
While tooth cleaning preparations steadily improved over the years, it was not until the early 1800s—when the modern toothpaste was developed—that truly effective oral products became available. The first mouthwashes were basically solutions of grain alcohol and were likely developed accidentally during this era. One of the most famous brands, Listerine, was developed during the 1880s and is still sold today.
The antibacterial effect of fluoride was an important discovery for the development of modern mouthwashes. In the early 1900s a dentist named Frederick McKay found that some of his patients had a condition called mottled enamel. He found that this condition was linked to a reduction in tooth decay. In 1931, he tested the drinking water that these patients consumed and found a high level of natural fluoride. By the early 1940s, other workers had determined that fluoride in drinking water at one part per million would reduce tooth decay without causing mottling. Various testing went on during the rest of this decade and by the 1950s it was recommended by the United States federal government that all public water sources be fortified with fluoride. This discovery led to the development of toothpastes and mouthwashes that contained fluoride compounds. During the years that followed, various raw materials have been developed that have an antibacterial effect but do not contain alcohol. Additionally, materials that prevent tartar, whiten teeth, and reduce cavities have also been discovered and added to mouthwash formulas.
Raw Materials
Mouthwashes are generally composed of diluents, antibacterial agents, soaps, flavorings, and colorants. The primary ingredient in most mouthwashes is water, a diluent, making up over 50% of the entire formula. The water is specially treated to remove various particles and ions which might impact flavor. Water treated as such is called deionized water. The sources of water vary, coming from reserves such as underground wells, lakes, and rivers. Alcohol is another diluent typically used in up to 20% of the formula. While early mouthwashes used alcohol extensively; today its use is limited because of governmental regulations and consumer desires.
Numerous antibacterial agents have been employed in mouthwash formulations. These include ingredients like phenols, thymol, salol, tannic acid, hexachloraphene, chlorinated thymols, and quaternary ammonium compounds. Chlorinated phenols like parachlormetacresol have both an antibacterial effect and a desired flavor. Thymol, which is obtained from volatile oils, is used at low concentrations and in conjunction with other ingredients. Hexachloraphene is substantive to the mucous membrane which makes it ideal for longer lasting formulas. Quatemary compounds are often used because of their non-toxic and non-irritant nature. They are effective against plaque.
Color and flavor are added to the formulas to improve the consumer acceptability of the mouthwash ingredients. Flavor is an essential feature of a mouthwash because it has the most consumer perceptible impact. In the United States, flavorants such as peppermint, menthol, methyl salicylate, and eugenol are commonly used. The most common colors, blue and green, are the result of adding governmentally approved and certified FD&C dyes. Some mouthwash formulas also include a synthetic detergent to give extra foaming and cleansing action.
Design
In general there are three types of mouthwashes. There are antibacterial products that reduce the bacterial population of the mouth. These products have a fresh taste and improve breath odor. The second type are fluoride mouthwashes, which help to improve the fluoride layer on tooth enamel. Finally, there are remineralizing mouthwashes that help repair various lesions in the mouth.
Mouthwashes are sold in a variety of flavors and colors. The most popular is the golden colored, medicinal-tasting Listerine. Blue or green mint varieties are also common. There are mouthwash products that are geared specifically toward smokers. These products are designed to remove tobacco tar stains as well as freshen breath and whiten teeth. There are also tartar control mouthwashes and those that kill the germs responsible for gingivitis and plaque.
The packaging for mouthwashes is typically a clear, plastic bottle. Since the products generally contain alcohol, child resistance and tamper-evidence closures are typically used. Additionally, some mouthwashes have a dosing feature which lets the consumer squeeze out an exact amount each time it is used.
The Manufacturing Process
After a mouthwash formula is designed, it is tested to ensure that minimal changes will occur over time regardless of the storage conditions. This testing, called stability testing, helps detect physical changes in such things as color, odor, and flavor. It can also provide information about product performance over time. In the United States, the Food and Drug Administration (FDA) requires that specific stability testing be done to ensure product performance during long term storage.
In general, the process for creating a mouthwash occurs in two distinct steps. First, a large batch of mouthwash is made then it is filled in the appropriate packaging.
Compounding
-
1 Mouthwash is made via a batch process in an area of the manufacturing
plant called compounding. Here operators, called compounders,
 Top Mouthwash Products Leaders
Top Mouthwash Products Leaders
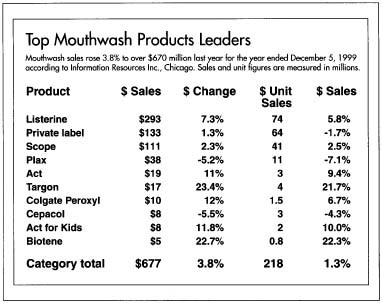
Mouthwash sales rose 3.8% to over $670 million last year fpr the year ended December 5, 1999 according to information Resources Inc., Chicago. Sales and unit figures are measured in millions.
make batches of 2,000-3,000 gal (7,571-11,356 L) of mouthwash following specific formula instructions. The raw materials are delivered to the compounding area by fork lift trucks. Compounders add them to the main batch tank where they are thoroughly mixed. Depending on the formula instructions, the batch is heated and cooled to get the raw materials to rapidly combine.Product $ Sales $ Change $ Unit Sales $ Sales Listerine $293 7.3% 74 5.8% Private label $133 1.3% 64 -1.7% Scope $111 2.3% 41 2.5% Plax $38 -5.2% 11 -7.1% Act $19 11% 3 9.4% Targon $17 23.4% 4 21.7% Colgate Peroxyl $10 12% 1.5 6.7% Cepacol $8 -5.5% 3 -4.3% Act for Kids $8 11.8% 2 10.0% Biotene $5 22.7% 0.8 22.3% Category total $677 3.8% 218 1.3% - 2 Materials which are used in large quantities—such as alcohol or water—are then pumped directly into the tank. This is done by simply setting computer controls to the appropriate amount and pushing a button. Computers also control the mixing speed and temperature of the batch. Depending on the size of the batch and the number of raw materials, a mouthwash can take anywhere from one to three hours to make.
Batch analysis
- 3 When the batch is completed, a sample is taken to the Quality Control (QC) lab. The appearance and flavor of the batch is examined to ensure that it meets the specifications laid out in the formula. QC chemists may also run pH determinations and viscosity checks. If some characteristic of the batch is found to be out of a specified range, adjustments may be made at this point. For example, colors can be modified by adding more dye.
- 4 After the batch is approved, it is pumped from the main tank to a holding tank. This holding tank may be directly hooked up to the filling lines where the product is put into individual packaging.
Filling
- 5 At the beginning of the filling line there is a large bin called a hopper which contains the empty bottles that will be filled. In this bin, the bottles are physically manipulated so that they come out standing upright on a conveyor belt. They are then moved to the filling carousel which contains the bulk mouthwash product.
- 6 The filling carousel has a series of piston filling heads that are designed to deliver an exact amount of mouthwash. As the bottles move around the carousel, the piston moves down and the mouthwash product is dispensed into the bottle.
- 7 After the bottles are filled, they are sent on a conveyor belt to a capping machine. The caps are also held in a large bin and correctly aligned. As the bottles pass the capping hopper, the caps are put on and either twisted or pushed in place.
- 8 From the capping station, the bottles are moved to a labeling machine. The labels are held on large spools and threaded through the machine. As the bottles pass by, the label is either stuck on using an adhesive or heat pressed.
- 9 Beyond labeling, the bottles are next moved to a boxing station. They are typically gathered in a group of 12 or 24 and dropped into a box. The boxes then move to a palleting machine and stacked. The pallets are moved via fork lifts to large trucks and shipped to distributors. High speed production lines like these can produce over 20,000 bottles per hour.
Quality Control
While quality control is a critical step in the batching process, it is also done at other points during manufacture. Workers are stationed at various points on the filling lines to inspect the production process. They examine things such as bottle quality, fill levels, and label placement. They also make sure that all the caps are put on correctly. Microbial contamination is also routinely checked during the filling process. Additionally, the packaging is checked for things such as bottle thickness, appearance, and weight to make sure the final product has the desired characteristics.
The Future
With advances in chemical technology, mouthwashes of the future will be designed with a larger array of and more improved functions. In the past, mouthwashes were primarily powerful breath fresheners. They eventually evolved into tooth protectors. Today, products are available to not only fight bad breath but whiten teeth and help battle cavity formation and gum recession.
Some new technologies that will undoubtedly be adapted to mouthwash products have recently been discovered. For example, researchers have found a peptide known as p1025, which can bond to the teeth and prevent the growth of naturally occurring bacteria. This prevents the cavity-causing bacteria to adhere to the tooth and thus inhibits cavity formation. Using this technology, they have created a mouthwash that may prevent tooth decay for up to three months.
Another new mouthwash may actually contain a good bacteria to kill the odor-and cavity-causing germ Streptococcus mutans. Using genetic engineering, scientists at the University of Florida College of Dentistry developed this bacterium and are now testing it in humans to determine whether it can be used. Ultimately, this new bacteria may be added to mouthwash products and thereby revolutionizing oral care.
Where to Learn More
Periodicals
Hickey, James. "Oral Care Market." Happi (February 2000). http://www.happi.com/special/febOO2.htm (January 2001).
Hickey, James. "Total Domination." Happi (February 1999). http://www.happi.com (January 2001).
Shaw, Anita. "Oral Care Market is All Smiles." Soap & Cosmetics 75, no.6 (June 1999): 28.
Travis, John. "Wash That Mouth Out With Bacteria!" Science News 157, no.12 (March 18, 2000): 190.
Wilson, Jim. "Mouthwash Cancels Cavities." Popular Mechanics 177, no.2 (February 2000): 15.
— Perry Romanowski
Comment about this article, ask questions, or add new information about this topic: