Amber
Background
Although considered a gem, amber is a wholly-organic material derived from the resin of extinct species of trees. In the dense forests of the Middle Cretaceous and Tertiary periods, between 10 and 100 million years ago, these resin-bearing trees fell and were carried by rivers to coastal regions. There, the trees and their resins became covered with sediment, and over millions of years the resin hardened into amber. Although many amber deposits remain in ocean residue, geological events often repositioned the amber elsewhere.
For thousands of years, amber has been carved and worked into beads, jewelry, and other types of ornamentation. However, today amber is valued primarily for the astounding array of fossils preserved inside. As sticky resin was exuded by the trees, animals, minerals, and plant materials were trapped in it. As the resin hardened, these fossils—called inclusions—were perfectly preserved, providing modern scientists with invaluable information about extinct species.
Unlike other types of fossils, amber fossils are three-dimensional, with life-like colors and patterns. Even the internal structures of cells may be intact. Often, insects were caught by the resin in active poses, along with their predators, prey, and internal and external parasites. Previously-unknown genera of fossilized insects have been discovered in amber. Intact frogs and lizards, snake skins, bird feathers, hair and bones of mammals, and various plant materials have been preserved in amber. In some cases, deoxyribonucleic acid (DNA) can be extracted from the fossilized organisms and compared with that of its modern-day counterparts.
History
Amber has been a highly-valued material since earliest times. Worked amber dating back to 11,000 B.C. has been found at archeological sites in England. Amber was widely believed to have magical healing powers. It was used to make varnish as long ago as 250 B.C. , and powdered amber was valued as incense. Amber was also traded throughout the world. By identifying the type of amber used in ancient artifacts, scholars can determine the geographical source of the amber and draw conclusions about early trade routes.
In about 600 B.C. , the Greek philosopher Thales rubbed amber with silk, causing it to attract dust and feathers. This static electricity was believed to be a unique property of amber until the sixteenth century, when English scientist William Gilbert demonstrated that it was characteristic of numerous materials. He called it electrification, after elektron, the Greek word for amber.
In the Western Hemisphere, the Aztecs and Mayans carved amber and burned it as incense. The Taino Indians of the island of Hispaniola offered gifts of amber to Christopher Columbus.
The decorative use of amber culminated in 1712 with the completion of an entire banquet room made of amber panels constructed for King Frederick I of Prussia. In the nineteenth century amber attained new significance when German scientists began studying the fossils imbedded in it.
Raw Materials
Resins are complex substances that include oily compounds called terpenes. Over time, some terpenes evaporate while others condense and become cross-linked to each other, forming hard polymers. However, different species of trees produce different types and amounts of resins. The exact structure and composition of amber depends on the makeup of the original tree resin, the age of the amber, the environment in which it was deposited, and the thermal conditions and geological forces to which it was exposed. Thus, even amber obtained from similar locations may vary in chemical structure and physical characteristics.
Types of amber
Although deposits of amber occur throughout the world, amber from the coast of the Baltic Sea is the best-known. It is called succinite amber because it contains a substantial amount of succinic acid. Most Baltic amber came from pine tree resin. Amber that lacks succinic acid is classified as retinite amber.
Amber from Mexico and the Dominican Republic began forming 20-30 million years ago from the resins of extinct species of Hymenaea or algarrobo trees. These flowering trees thrived in the canopy of extensive tropical rain forests. They produced copious amounts of resin that eventually hardened into amber. Torrential rains washed the amber to deltas where it was covered with silt. As sea levels changed, the amber settled on the sea floor and the sediment over it hardened into rock. Later, mountain formation pushed up the rocks.
Design
Physical characteristics
Many components of amber are similar to those of modern resins. However the cross-linking of these compounds makes the amber hard, with a high melting point and low solubility. Amber has a hardness of 2-3 on Mohs's scale, the standard for minerals and gems. On this scale, talc is I and diamond is 10. Amber softens at 302°F (150°C) and melts at 482-662°F (250-350°C). With a specific gravity of 1.05-1.12, amber is only slightly more dense than water. It will not completely dissolve in organic solvents.
Amber usually occurs as small irregular masses, nodules, or droplets. Although it can be many different colors, it is most often pale to golden yellow or orange and can be fluorescent. After a few years of exposure to light and air, amber often turns dark red and develops numerous cracks on the surface. Some amber is translucent or even transparent. However, trapped air bubbles can cause amber to be cloudy or opaque. Amber is a poor conductor of heat and large changes in temperature can cause it to fracture.
The Manufacturing
Process
Amber is extracted in different ways, depending on its location. Baltic amber washes up along the shores of the Baltic Sea and as far away as Denmark, Norway, and England. The largest deposits of North American amber are found on the surface of openpit clay mines in Arkansas. In New Jersey, Cretaceous amber is dug from the sand and clay of abandoned pit mines. It is screened, washed, and examined for inclusions. In Asia, amber is found in coal mines. Until the mid-twentieth century, highly-prized amber was mined from deep pits in northern Burma (now Myanmar).
Mining and washing
-
1 Drops or blocks of Baltic amber are mined from open pits of 40-60
million-year-old glauconite sand. Glauconite is a hydrated
potassium-iron silicate mineral and these deposits are called
"blue earth" because of their blue-green color. After the
surface has been cleared, the blue earth is dug out with steam shovels
and dredges. It is poured through grates at a washing plant, where
streams of water are used to separate the amber from the sand. In the
early twentieth century, up to one million lb (450,000 kg) of amber per
year were extracted from the blue earth layer of the Samland Peninsula
in the eastern Baltic.
Mexican and Dominican amber may be exposed by landslides on steep mountain slopes and extracted with picks and shovels. It also is mined from pits dug deep into the ground. Much Dominican amber is mined from narrow tunnels carved as far as 600 ft (183 m) into the sides of mountains. Water is baled or pumped out of the tunnels and the miners crawl through, chisel at the rock, and pick out the exposed amber. Dominican
amber is washed by the miner, sorted by size, and examined for inclusions.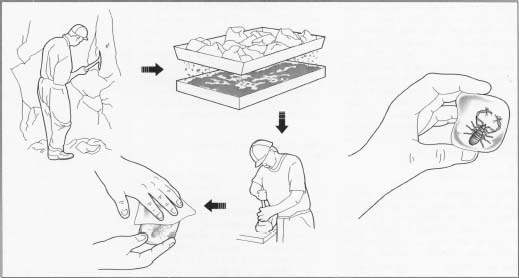 Baltic amber is mined and clarified to obtain a clear view of the inclusion.
Baltic amber is mined and clarified to obtain a clear view of the inclusion.
Clarifying and coloring
- 2 Large trapped air bubbles result in a foamy or frothy type of amber. Microscopic bubbles result in bony or osseous amber that looks like dried bone. Very cloudy amber is called bastard. Amber is clarified by heating in rapeseed oil. The oil penetrates bubbles near the surface and reduces the cloudiness, making even bony or bastard amber more transparent. Amber also may be clarified by heating under pressure with nitrogen and then baking in an oven. Clarification darkens the amber and produces disc-like stress marks, called "sun spangles." Amber may be stained green or reddish. Mexican and Dominican amber is usually clear and transparent and does not need clarifying.
Cutting and reshaping
- 3 For jewelry or carving, amber usually is worked by hand, with a jeweler's saw and fine-toothed files. It is wet-sanded with 320-grit cloth and finished with a 400- or 600-grit wet-sanding cloth. It can be drilled with dry steel drills, using a low speed and slight pressure, to prevent heating and cracking.
- 4 To obtain a clear view of inclusions, one end of an amber piece may be chipped off. Amber with inclusions may be cut or reshaped for examination of the biological specimen or to separate two specimens. Cutting is done with a jeweler's hand saw or, for larger pieces, with a high-speed trim saw with a diamond blade, at speeds up to 4,200 rpm.
- 5 Reshaping is done with various grades of sandpaper. Rough edges from the saw blade may be smoothed with 200- and 400-grit paper, by hand or with a belt sander equipped with a water cooling system, to remove dust and prevent overheating and fracturing or glazing.
Polishing
-
6 Amber for jewelry is polished with tin oxide or cerium oxide, using a
leather buff, Jelt wheel or pad, or chamois board. Periodic polishing
with a silicone-based wax restores shine and decreases evaporation and
surface oxidation.
Dominican amber is polished with a sander, following the natural contours. Surface oxidation of Dominican amber diminishes the fluorescence and the blue, green, or purple color. Removing the outer layer and repolishing restores the fluorescence. Repolishing may be done by hand or with a cotton buffing wheel, using dental polishing compound, an abrasive for plastics, or other fine neutral-colored polishing compounds. A final hand polishing removes the polishing compound.
Cretaceous amber more than 65 million years old is very brittle and fractured. After several years of exposure, it is prone to disintegration. Encasing Cretaceous amber in a synthetic resin helps to preserve it.
Pressing
- 7 For producing gems, small clear pieces of amber are softened and fused in a vacuum with steam at 400°F (204°C) or above.
- 8 The pieces are pressed through a fine steel sieve or mesh, mixed, and hardened into blocks. This pressed amber is called ambroid or amberoid and may contain bubbles that have elongated under the heat and pressure. Sometimes modern insect inclusions are inserted into pressed amber and the ambroid may be dyed, usually dark red.
Other processing
- 9 Small pieces of poor quality amber, including about 90% of Baltic amber, are distilled in huge, dry iron retorts. About 60% is recovered as amber colophony, a high-grade varnish. Another 15—20% becomes amber oil, used in medicines, casting, and the highest grade of varnish. About 2%of the products are distilled acids, such as succinic acid, that are used for medicines and varnishes.
Quality Control
Harder and, presumably, older amber is usually considered to be of higher quality. Since mining costs are 28% higher than the value of raw amber, its value is based primarily on its inclusions or on its eventual processing into jewelry and art objects. Therefore, amber is graded according to its size and beauty, as well as the presence and type of inclusions.
Imitation amber with fake inclusions has been produced for at least 600 years. Fresh resins, synthetic polystyrenes, Bakelite, epoxy resins, celluloid, colored glass, plastics, and polyesters all have been used for imitation amber. However, true amber can be distinguished by its hardness, melting temperature, lack of solubility, fluorescence, specific gravity, refractive index (measure of the degree that it bends light), and odor on burning. Sometimes true amber is fractured, a cavity is carved in it for an inclusion embedded in fresh resin, and the piece is resealed.
Byproducts/Waste
About 90% of the world's extractable amber is located in the Kaliningrad region of Russia on the Baltic Sea. There, amber mining and processing has caused widespread environmental degradation. More than 100 million tons of waste has been discharged into the Baltic from the Palmnicken (Yantarny) mine over the past century. This insoluble waste causes high turbidity in the Baltic Sea. The waters of the pollution-sensitive Baltic take 25-30 years to renew themselves.
The Future
The easily-extracted, top layers of Baltic amber were exhausted by the mid-1800s. However it is estimated that over 180,000 tons of amber remain in the Yantarny mine in Kaliningrad. At the current rate of extracetion, amber could be mined there for another 300 years. In addition, mining has resumed in Myanmar and the high-quality Burmese amber is being sold to museums.
Although the process of amber formation from tree resin continues, it takes millions of years for the resin to harden into amber. As amber deposits are depleted by mining, and resin-bearing trees are cut or burned rather than allowed to fossilize, the supply of raw amber will continue to dwindle.
Where to Learn More
Books
Anderson, K. B., and J. C. Crelling, eds. Amber, Resinite, and Fossil Resins. Washington, DC: American Chemical Society, 1995.
Grimaldi, David A. Amber: Window to the Past. New York: Harry N. Abrams, Inc. and the American Museum of Natural History, 1996.
Poinar Jr., George, and Roberta Poinar. The Amber Forest: A Reconstruction of a Vanished World. Princeton: Princeton University Press, 1999.
Other
"Amber Trade and the Environment in the Kaliningrad Oblast." TED Case Studies. 27 July 2001. < http://gurukul.ucc.american.edu/ted/amber.htm >.
Brost, Leif. "Amber: A Fossilized Tree Resin." The Swedish Amber Museum Home Page. 27 July 2001. < http://www.brost.se/eng/education/facts.html >.
Margaret Alic
Comment about this article, ask questions, or add new information about this topic: