Polyester
Background
Polyester is a synthetic fiber derived from coal, air, water, and petroleum. Developed in a 20th-century laboratory, polyester fibers are formed from a chemical reaction between an acid and alcohol. In this reaction, two or more molecules combine to make a large molecule whose structure repeats throughout its length. Polyester fibers can form very iong molecules that are very stable and strong.
Polyester is used in the manufacture of many products, including clothing, home furnishings, industrial fabrics, computer and recording tapes, and electrical insulation. Polyester has several advantages over traditional fabrics such as cotton. It does not absorb moisture, but does absorb oil; this quality makes polyester the perfect fabric for the application of water-, soil-, and fire-resistant finishes. Its low absorbency also makes it naturally resistant to stains. Polyester clothing can be preshrunk in the finishing process, and thereafter the fabric resists shrinking and will not stretch out of shape. The fabric is easily dyeable, and not damaged by mildew. Textured polyester fibers are an effective, nonallergenic insulator, so the material is used for filling pillows, quilting, outerwear, and sleeping bags.
History
In 1926, United States-based E.I. du Pont de Nemours and Co. began research into very large molecules and synthetic fibers. This early research, headed by W.H. Carothers, centered on what became nylon, the first synthetic fiber. Soon after, in the years 1939-41, British research chemists took interest in the du Pont studies and conducted their own research in the laboratories of Calico Printers Association, Ltd. This work resulted in the creation of the polyester fiber known in England as Terylene.
In 1946, du Pont purchased the right to produce this polyester fiber in the United States. The company conducted some further developmental work, and in 1951, began to market the fiber under the name Dacron. During the ensuing years, several companies became interested in polyester fibers and produced their own versions of the product for different uses. Today, there are two primary types of polyester, PET (polyethylene terephthalate) and PCDT (poly-1, 4-cyclohexylene-dimethylene terephthalate). PET, the more popular type, is applicable to a wider variety of uses. It is stronger than PCDT, though PCDT is more elastic and resilient. PCDT is suited to the heavier consumer uses, such as draperies and furniture coverings. PET can be used alone or blended with other fabrics to make clothing that is wrinkle and stain resistant and retains its shape.
Raw Materials
Polyester is a chemical term which can be broken into poly, meaning many, and ester, a basic organic chemical compound. The principle ingredient used in the manufacture of polyester is ethylene, which is derived from petroleum. In this process, ethylene is the polymer, the chemical building block of polyester, and the chemical process that produces the finished polyester is called polymerization.
The Manufacturing
Process
Polyester is manufactured by one of several methods. The one used depends on the form the finished polyester will take. The four basic forms are filament, staple, tow, and fiberfill. In the filament form, each individual strand of polyester fiber is continuous in length, producing smooth-surfaced fabrics. In staple form, filaments are cut to short, predetermined lengths. In this form polyester is easier to blend with other fibers. Tow is a form in which continuous filaments are drawn loosely together. Fiberfill is the voluminous form used in the manufacture of quilts, pillows, and outerwear. The two forms used most frequently are filament and staple.
Manufacturing Filament Yarn
Polymerization
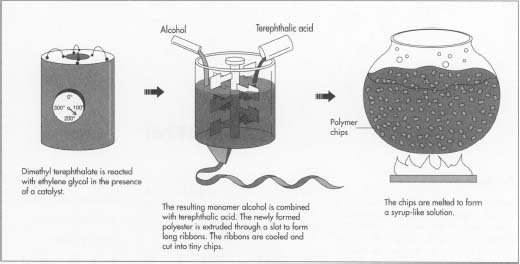
- 1 To form polyester, dimethyl terephthalate is first reacted with ethylene glycol in the presence of a catalyst at a temperature of 302-410°F (150-210°C).
- 2 The resulting chemical, a monomer (single, non-repeating molecule) alcohol, is combined with terephthalic acid and raised to a temperature of 472°F (280°C). Newly-formed polyester, which is clear and molten, is extruded through a slot to form long ribbons.
Drying
- 3 After the polyester emerges from polymerization, the long molten ribbons are allowed to cool until they become brittle. The material is cut into tiny chips and completely dried to prevent irregularities in consistency.
Melt spinning
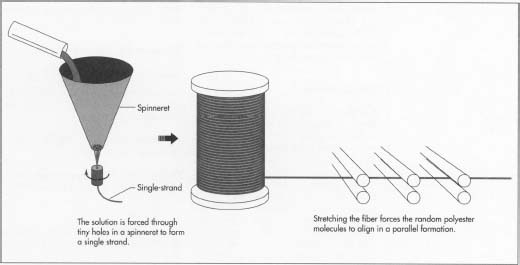
- 4 Polymer chips are melted at 500-518°F (260-270°C) to form a syrup-like solution. The solution is put in a metal container called a spinneret and forced through its tiny holes, which are usually round, but may be pentagonal or any other shape to produce special fibers. The number of holes in the spinneret determines the size of the yarn, as the emerging fibers are brought together to form a single strand.
- 5 At the spinning stage, other chemicals may be added to the solution to make the resulting material flame retardant, antistatic, or easier to dye.
Drawing the fiber
- 6 When polyester emerges from the spinneret, it is soft and easily elongated up to five times its original length. The stretching forces the random polyester molecules to align in a parallel formation. This increases the strength, tenacity, and resilience of the fiber. This time, when the filaments dry, the fibers become solid and strong instead of brittle.
- 7 Drawn fibers may vary greatly in diameter and length, depending on the characteristics desired of the finished material. Also, as the fibers are drawn, they may be textured or twisted to create softer or duller fabrics.
Winding
- 8 After the polyester yarn is drawn, it is wound on large bobbins or flat-wound packages, ready to be woven into material.
Manufacturing Staple Fiber
In making polyester staple fiber, polymerization, drying, and melt spinning (steps 1-4 above) are much the same as in the manufacture of filament yarn. However, in the melt spinning process, the spinneret has many more holes when the product is staple fiber. The rope-like bundles of polyester that emerge are called tow.
Drawing tow
- 1 Newly-formed tow is quickly cooled in cans that gather the thick fibers. Several lengths of tow are gathered and then drawn on heated rollers to three or four times their original length.
Crimping
- 2 Drawn tow is then fed into compression boxes, which force the fibers to fold like an accordion, at a rate of 9-15 crimps per inch (3-6 per cm). This process helps the fiber hold together during the later manufacturing stages.
Setting
- 3 After the tow is crimped, it is heated at 212-302°F (100-150°C) to completely dry the fibers and set the crimp. Some of the crimp will unavoidably be pulled out of the fibers during the following processes.
Cutting
- 4 Following heat setting, tow is cut into shorter lengths. Polyester that will be blended with cotton is cut in 1.25-1.50 inch (3.2-3.8 cm) pieces; for rayon blends, 2 inch (5 cm) lengths are cut. For heavier fabrics, such as carpet, polyester filaments are cut into 6 inch (15 cm) lengths.
The Future
Following its introduction to the United States in 1951, polyester quickly became the country's fastest-growing fiber. Easy care of the permanent press fabric made polyester doubleknits extremely popular in the late 1960s. However, polyester has suffered an "image problem" since that time, and clothes made out of polyester were often devalued and even ridiculed. Several new forms of polyester introduced in the early 1990s may help revitalize the image of polyester. A new form of polyester fiber, called microfiber, was introduced to the public in 1991. More luxurious and versatile than traditional polyester, microfiber fabrics are difficult to tell apart from silk fabrics. Clothing designers such as Mary McFadden have created a line of clothes using this new form of polyester. Textile researchers at North Carolina State University are developing a form of polyester that may be as strong as Kevlar, a superfiber material used to make bulletproof vests. This type of polyester may eventually be used as composite materials for cars and airplanes.
Where To Learn More
Books
Corbman, Bernard P. Textiles: Fiber to Fabric. 6th ed. Gregg Division, McGraw-Hill, 1983, pp. 374-92.
Encyclopedia of Textiles. 3rd ed. Prentice-Hall, Inc., 1980, pp. 28-33.
Polyester: Fifty Years of Achievement. State Mutual Book & Periodical Service, 1993.
Periodicals
Fellingham, Christine. "Will You Learn to Love Polyester?" Glamour, April 1992, p. 204.
Templeton, Fleur. "Show Me a Bulletproof Leisure Suit, In Pink." Business Week, July 6, 1992, p. 65.
Thomas, Marita. "At 50 Years, Polyester Gains New Fashion Vitality." Textile World, December 1993, p. 62+.
— Kristine M. Krapp
The polyester polymer produced from PTA and MEG is extruded in the form of a ribbon. This ribbon is then converted into chips.
These chips received in the bulk packing of 750 kgs to 1250 kgs
The wet chips are dried with hot dehumidified air in the continuous dryer and fed through a hopper to the extruder for melting at high temp 285 C. This molten polymer is then extruded under high pressure through spinnerettes down to cylinder. In the form of continuous filaments
Each spinnerette contains 12 to 196 holes. A cool controlled air is blowing across the bunch of filaments for proper cooling. A spinning finish is applied at this stage as a lubricant and an antistatic agent. The yarn is partially drawn between the spinnerettes and the spin finish applicator before it is wind at very high speed on winders in the form of cheeses.
.
the yarn produced is deoffed from the winders checked for the properties and packed in the pallets of 60 spools each.
first yarn is drawn after oil finishing and befor winding,between spineret and spin finish applicator there isnt any drawing area
second oil finishing is not just for anti statin effect,it is mainly for sticking filaments together
My question is that how much ammount of oil content is present in the automotive greige polyester fabric ...