Indigo
Background
Indigo, or indigotin, is a dyestuff originally extracted from the varieties of the indigo and woad plants. Indigo was known throughout the ancient world for its ability to color fabrics a deep blue. Egyptian artifacts suggest that indigo was employed as early as 1600 B.C. and it has been found in Africa, India, Indonesia, and China.
The dye imparts a brilliant blue hue to fabric. In the dying process, cotton and linen threads are usually soaked and dried 15-20 times. By comparison, silk threads must be died over 40 times. After dying, the yarn may be sun dried to deepen the color. Indigo is unique in its ability to impart surface color while only partially penetrating fibers. When yarn died with indigo is untwisted, it can be seen that the inner layers remain uncolored. The dye also fades to give a characteristic wom look and for this reason it is commonly used to color denim. Originally extracted from plants, today indigo is synthetically produced on an industrial scale. It is most commonly sold as either a 100% powder or as a 20% solution. Through the early 1990s, indigo prices ranged near $44/lb ($20/kg).
History
The name indigo comes from the Roman term indicum, which means a product of India. This is somewhat of a misnomer since the plant is grown in many areas of the world, including Asia, Java, Japan, and Central America. Another ancient term for the dye is nil from which the Arabic term for blue, al-nil, is derived. The English word aniline comes from the same source.
The dye can be extracted from several plants, but historically the indigo plant was the most commonly used because it is was more widely available. It belongs to the legume family and over three hundred species have been identified. Indigo tinctoria and I. suifruticosa are the most common. In ancient times, indigo was a precious commodity because plant leaves contain only about small amount of the dye (about 2-4%). Therefore, a large number of plants are required to produce a significant quantity of dye. Indigo plantations were founded in many parts of the world to ensure a controlled supply.
Demand for indigo dramatically increased during the industrial revolution, in part due to the popularity of Levi Strauss's blue denim jeans. The natural extraction process was expensive and could not produce the mass quantities required for the burgeoning garment industry. So chemists began searching for synthetic methods of producing the dye. In 1883 Adolf von Baeyer (of Baeyer aspirin fame) researched indigo's chemical structure. He found that he could treat omega-bromoacetanilide with an alkali (a substance that is high in pH) to produce oxindole. Later, based on this observation, K. Heumann identified a synthesis pathway to produce indigo. Within 14 years their work resulted in the first commercial production of the synthetic dye. In 1905 Baeyer was awarded the Nobel Prize for his discovery.
At the end of the 1990s, the German based company BASF AG was the world's leading producer, accounting for nearly 50% of all indigo dyestuffs sold. In recent years, the synthetic process used to produce indigo has come under scrutiny because of the harsh chemicals involved. New, more environmentally responsible methods are being sought by manufacturers.
Raw Materials
The raw materials used in the natural production of indigo are leaves from a variety of plant species including indigo, woad, and polygonum. Only the leaves are used since they contain the greatest concentration of dye molecules. In the synthetic process, a number of chemicals are employed as described below.
The Manufacturing Process
Natural extraction
- 1 Plant extraction of indigo requires several steps because the dye itself does not actually exist in nature. The chemical found in plant leaves is really indican, a precursor to indigo. The ancient process to extract indican from plant leaves and convert it to indigo has remained unchanged for thousands of years. In this process, a series of tanks are arranged in a step wise fashion. The upper-most tank is a fermentation vessel into which the freshly cut plants are placed. An enzyme known as indimulsin is added to hydrolyze, or break down, the indican into indoxyl and glucose. During this process carbon dioxide is given off and the broth in the tank turns a murky yellow.
-
2 After about 14 hours, the resulting liquid is drained into a second tank. Here, the indoxyl-rich mixture is stirred with paddles to mix it with air. This allows the air to oxidize the indoxyl to indigotin, which settles to the bottom of the tank. The upper layer of liquid is siphoned away and the settled pigment is transferred to a third tank where it is heated to stop the fermentation process. The resultant mixture is filtered to remove impurities and dried to form a thick paste.
Historically, the Japanese have used another method which involves extracting indigo from the polygonum plant. In this process the plant is mixed with wheat husk powder, limestone powder, lye ash, and sake. The mixture is allowed to ferment for about one week to form the dye pigment which is called sukumo.
Synthetic production
- 3 A variety of synthetic chemical processes have been used to produce indigo. All these processes involve combining a series of chemical reactants under controlled conditions. The reactants undergo a series of reactions which result in the formation of the indigo molecule. A number of other chemical byproducts are also produced in this reaction.
- 4 These synthesis reactions are conducted in large stainless steel or glass reaction vessels. These vessels are equipped with jackets to allow steam or cold water to flow around the batch as the reactions progress. Because of the complexity of these chemical processes, the dye is usually made in batch quantities. There are, however, a few methods invented by the Germans for continuous process manufacturing.
Types of reactions
- 5 The first commercial method of producing indigo was based on Heumann's work. In this method, N-phenylglycine is treated with alkali to produce indoxyl, which can be converted to indigotin by contact with air. However, the amount of dye yielded by this process is very low. Another, more efficient, synthesis route utilizes anthranilic acid. This process was popular with major manufacturers, such as BASF and Hoechst, for over 30 years. A variation of this method (which has become widely used) involves the reaction of aniline, formaldehyde, and hydrogen cyanide to form phenylglycinonitrile. This material is then hydrolyzed to yield phenylglycine which is then converted to indigotin. Currently, a method which uses sodamide with alkali to convert phenylglycine to indoxyl. Sodamide reacts with excess water, thus lowering the overall reaction temperature from almost 570°F (300°C) to 392°F (200°C). This results in a much more efficient reaction process.
Finishing operations
-
6 After the chemical reaction process is complete, the finished dye must be washed to remove impurities and then dried. The dried powder can be packed in drums or reconstituted with water to form a 20% solution and filled in pails.
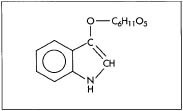 The chemical symbol for indican, the compound found in the leaves of the indigo plant that is used to make indigo dye.
The chemical symbol for indican, the compound found in the leaves of the indigo plant that is used to make indigo dye.
Quality Control
During indigo manufacture, the reaction process is continuously monitored to ensure the chemicals are combined in the proper ratios. Key elements that must be controlled include the pH (or acid/base quality of the batch), the temperature (which controls the speed of the reaction), and the reaction time (which determines the degree of completion). If any of these variables deviate from specifications, the resulting reaction product can be affected. Typically, poor quality control results in lower yield of the dye, which increases costs for the manufacturer.
To ensure that manufacturers can consistently purchase the same shade of dye, indigo is assigned a Color Index number that defines its shade. It is designated as "CI Natural Blue CI 75780."
Byproducts/Waste
Indigo production produces a variety of waste products which must be handled carefully. In addition to the reactants described above, there are other reaction side products that are produced along with the indigo. Some of these materials are considered to be hazardous and must be disposed of in accordance with local and federal chemical waste disposal guidelines. These waste chemicals can enter the environment in at least three different ways. The first is during the actual manufacture of the molecule. The second is when the dye is applied to the yarn, and the third is when the dye is eluted into the wash water during the initial stonewashing or wet processing of the fabric. This last route typically occurs during the production of denim fabric.
The Future
Much of the need for indigo is being met with other types of blue dyes and today most of the indigo used by the world is made out-side the United States. Researchers are concentrating on new methods of indigo manufacture that are more environmentally friendly. One promising future method involves using biocatalysts in the dye reaction process. Indigo dye may be one of the first high-volume chemicals made through a biological route. Genencor International, of Rochester New York, is evaluating a process to produce indigo using biotechnology. According to Charles T. Goodhue, Genencor's Program Director/Biocatalysis Research and Development, indigo produced by this method is chemically the same as the regular synthetic dye and behaves identically in dyeing tests. However, at this time the technology is expensive and production costs could be prohibitive. Genencor is seeking a major market partner to work with them in the development of this new technology.
Manufacturers who use indigo in dying operations are also seeking to improve their use of the dye. For example, Burlington's Denim Division introduced a technology in 1994 they call "Stone Free," which allows indigo dye in the fabric to break down 50% faster in the stonewash cycle. Compared to traditional methods of stonewashing fabric dyed with indigo, their new process uses few, if any, pumice stones which help give the fabric its faded look. Therefore, pumice stone handling and storage costs are reduced, along with time required to separate pumice from garments after stonewashing. It also uses much less bleach. Therefore, this new process not only reduces garment damage, but also reduces waste produced by the stones and bleach.
Where to Learn More
Books
Kirk, R. E., and D. F. Othmer (ed.) Encyclopedia of Chemical Technology: Alkoxides, Metal to Antibiotics (Peptides). Wiley-Interscience, John Wiley and Sons, 1978.
Periodicals
Guilbaut, G. B., and D. W. Kramer. "Resorufin Butyrate and Indoxyl Acetate as Fluorogenic Substrates for Cholinesterases." Analytical Chemistry 37 (1965):120-23.
McCurry, John. "Burlington Debuts Stone Free Denim." Textile World 144, no.3 (March 1994): 120-123.
Rotman, David, and Emma Chynoweth. "The Quest for Reduced Emissions, Greener Processes." Chemical Week 153, no.1 (July 7,1993): 117.
— Randy Schueller