Aspartame
Background
Aspartame is an artificial sweetener used in reduced calorie foods. It is derived primarily from two naturally occurring amino acids chemically combined and designated by the chemical name N-L-aaspartyl-L-phenylalanine-l-methyl ester (APM). Discovered inadvertently in 1965, it was later patented and is currently the most utilized artificial sweetener in the United States.
Aspartame is a white, odorless, crystalline powder. It is about 200 times sweeter than sugar and is readily dissolvable in water. It has a sweet taste without the bitter chemical or metallic aftertaste reported in other artificial sweeteners. These properties make it a good ingredient to use as a sugar replacement in many food recipes. However, aspartame does tend to interact with other food flavors, so it cannot perfectly replace sugar. Recipes for baked goods, candies, and other products must be modified if aspartame is utilized. Although aspartame can be used in microwave recipes, it is sensitive to extensive heating, which makes it unsuitable for baking.
The fact that aspartame provides sweetness and flavor without imparting other physical characteristics such as bulk or calories like other sweeteners makes it unique. Another useful trait is that it has a synergistic effect with other sweeteners, making it possible to use less total sweetener. In addition to sweetening foods, aspartame is used to reduce calories, and intensify and extend fruit flavors.
History
Humans have desired foods with a sweet taste for thousands of years. Ancient cave paintings at Arana in Spain show a neolithic man taking honey from a wild bee's nest. It has been suggested that early humans might have used the sweet taste of foods to tell them which ones would be safe to eat. It is even thought that the desire for sweet taste might be an innate human trait. Unfortunately, many of the foods that are naturally sweet contain relatively large amounts of calories and carbohydrates.
Alternative sweeteners were developed to provide the sweet taste without the unnecessary calories. They also provide the additional benefits of enhancing the palatability of pharmaceuticals, aiding in the management of diabetes, and providing a cost-effective source where sugar is not available. The first one, saccharin, was discovered in 1879 and has been used in products such as toothpaste, mouthwash, and sugarless gum.
The sugarlike taste of aspartame was discovered accidentally by James Schlatter, an American drug researcher at G.D. Searle and Co. in 1965. While working on an antiulcer drug, he inadvertently spilled some APM on his hand. Figuring that the material was not toxic, he went about his work without washing it off. He discovered APM's sweet taste when he licked his finger to pick up a piece of weighing paper. This initial breakthrough then led the company to screen hundreds of modified versions of APM. However, none of these materials offered all of the advantages found in the original compound, including economical manufacturing, excellent taste quality and potency, natural metabolic pathways for digestion, excellent stability, and very low toxicity. Consequently, the company pursued and was granted United States patent 3,492,131 and various international patents, and the initial discovery was commercialized. The U.S. patent expired in 1992, and the technology is now available to any company who wants to use it.
After many years of toxicity testing, the FDA initially approved aspartame's use as a sweetener in 1980. However, a hallmark of synthetic chemicals used in food products is that their safety is under constant scrutiny. Aspartame is no exception and has been surrounded by some controversy concerning its safety since its introduction. Most of these concerns were put to rest in late 1984, when after investigating various aspartame-related complaints, the FDA and the Centers for Disease Control concluded that the substance is safe and does not represent a widespread health risk. This conclusion was further supported by the American Medical Association in 1985, and aspartame has been gaining market share ever since. In addition to its use in the United States, aspartame has also been approved for use in over 93 foreign countries.
Aspartame has been marketed since 1983 by Searle under the brand names NutraSweet' and Equal'. Currently, NutraSweet' is a very popular ingredient and is used in more than 4,000 products, including chewing gum, yogurt, diet soft drinks, fruit-juices, puddings, cereals, and powdered beverage mixes. In the U.S. alone, NutraSweet®'s sales topped $705 million in 1993, according to the company.
Raw Materials
Aspartame is primarily derived from compounds called amino acids. These are chemicals which are used by plants and animals to create proteins that are essential for life. Of the 20 naturally occurring amino acids, two of them, aspartic acid and phenylalanine, are used in the manufacture of aspartame.
All amino acids molecules have some common characteristics. They are composed of an amino group, a carboxyl group, and a side chain. The chemical nature of the side chain is what differentiates the various amino acids. Another characteristic of amino acids is the ability to form different molecular configurations known as isomers. These isomers are designated by the letters L and D. Aspartame is composed of only L, L isomers; none of the other isomer combinations taste sweet. The sweet taste of aspartame could not have been predicted by looking at the two amino acids that it is derived from. L-aspartic acid has a flat taste and L-phenylalanine tastes bitter. However, when the two compounds are chemically combined and the L-phenylalanine is slightly modified, a sweet taste is achieved.
Aspartic acid is one of five amino acids that have a "charged" side group. The charged side group on aspartic acid is (-CH 2 -COOH). When put in water, this material ionizes and becomes negatively charged. Phenylalanine has a nonpolar, hydrophobic side group which is not compatible with water. It is made up of a six carbon ring and is attached to the main amino acid backbone via a methyl (-CH 2 ) group. Prior to synthesis into aspartame, it is reacted with methanol. This adds a methyl group which is linked to the molecule by an oxygen, and the compound is converted to a methyl ester. The methanol required for the synthesis of aspartame has the chemical structure (CH 3 -OH). This is a very common material and is used extensively by organic chemists for various chemical syntheses.
The Manufacturing
Process
Although its components—aspartic acid, phenylalanine, and methanol—occur naturally in foods, aspartame itself does not and must be manufactured. NutraSweet' (aspartame) is made through fermentation and synthesis processes.
Fermentation
Direct fermentation produces the starting amino acids needed for the manufacture of aspartame. In this process, specific types of bacteria which have the ability to produce certain amino acids are raised in large quantities. Over the course of about three days, the amino acids are harvested and the bacteria are destroyed.
-
1 To start the fermentation process, a sample from a pure culture of
bacteria is put into a test tube containing the nutrients necessary for
its growth. After this initial inoculation the bacteria begin to
multiply. When their population is large enough, they are transferred to
a seed tank. The bacterial
strains used to make L-aspartic acid and L-phenylalanine are B. flavum and C. glutamicum respectively.
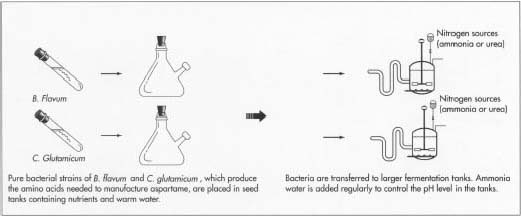
- 2 The seed tank provides an ideal environment for growing more bacteria. It is filled with the things bacteria need to thrive, including warm water and carbohydrate foods like cane molasses, glucose, or sucrose. It also has carbon sources like acetic acid, alcohols or hydrocarbons, and nitrogen sources such as liquid ammonia or urea. These are required for the bacteria to synthesize large quantities of the desired amino acid. Other growth factors such as vitamins, amino acids, and minor nutrients round out seed tank contents. The seed tank is equipped with a mixer, which keeps the growth medium moving, and a pump, which delivers filtered, compressed air. When enough bacterial growth is present, the contents from the seed tank are pumped to the fermentation tank.
- 3 The fermentation tank is essentially a larger version of the seed tank. It is filled with the same growth media found in the seed tank and also provides a perfect environment for bacterial growth. Here the bacteria are allowed to grow and produce large quantities of amino acids. Since pH control is vital for optimal growth, ammonia water is added to the tank as necessary.
- 4 When enough amino acid is present, the contents of the fermentation tank are transferred out so isolation can begin. This process starts with a centrifugal separator, which isolates a large portion of the bacterial amino acids. The desired amino acid is further segregated and purified in an ion-exchange column. From this column, the amino acids are pumped to a crystallizing tank and then to a crystal separator. They are then dried and readied for the synthesis phase of aspartame production.
Synthesis
Aspartame can be made by various synthetic chemical pathways. In general, phenylalanine is modified by a reaction with methanol and then combined with a slightly modified aspartic acid which eventually forms aspartame.
- 5 The amino acids derived from the fermentation process are initially modified to produce aspartame. Phenylalanine is reacted with methanol resulting in a compound called L-phenylalanine methyl ester. Aspartic acid is also modified in such a way to shield various portions of the molecule from the effects of further reactions. One method is by reacting the aspartic acid with substances that result in added benzyl rings to protect these sites. This ensures that further chemical reactions will occur only on specific parts of the aspartic acid molecule.
-
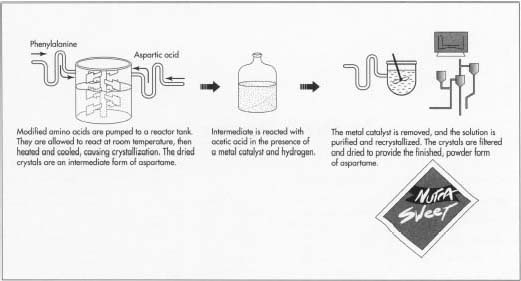
6 After the amino acids are appropriately modified, they are pumped into
a reactor tank, where they are allowed to mix at room temperature for 24
hours. The temperature
is then increased to approximately 149°F (65 °C) and maintained for another 24 hours. The reaction is then cooled to room temperature. It is diluted with an appropriate solvent and cooled to about 0°F (-18°C), causing crystallization. The crystals are then isolated by filtration and dried. These crystals are an intermediate of aspartame which must be further modified.
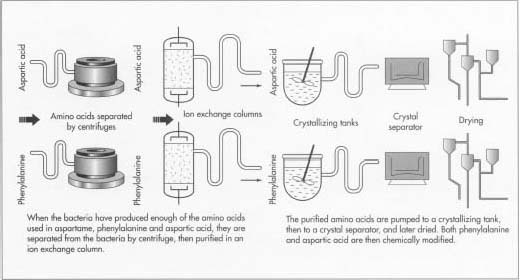
- 7 The intermediate is converted to aspartame by reacting it with acetic acid. This reaction is performed in a large tank filled with an aqueous acid solution, a palladium metal catalyst, and hydrogen. It is thoroughly mixed and allowed to react for about 12 hours.
Purification
- 8 The metal catalyst is removed by filtration, and the solvent is distilled, leaving a solid residue. This residue is purified by dissolving it in an aqueous ethanol solution and recrystallizing. These crystals are filtered and dried to provide the finished, powder aspartame.
Quality Control
The quality of the compounds is checked regularly during the manufacturing process. Of particular importance are frequent checks of the bacterial culture during fermentation. Also, various physical and chemical properties of the finished product are checked, such as pH level, melting point, and moisture content.
The Future
Currently, there are only three alternative sweeteners in the United States that can be used in food products. While aspartame is perhaps one of the best available, scientists are looking for new ways to make these sweeteners taste as much like sugar as possible. Their research has been focused in three areas, including finding new derivatives, blending sweeteners, and enhancing the efficiency of aspartame.
Most of the chemical derivative work has centered on finding compounds which will fit into the taste bud receptors better than traditional aspartame. Using aspartame as the model, researchers believe they will be able to improve various characteristics by making slight modifications. For example, they have found that when L-aspartic acid alone is modified in a certain way, it gives products that have a sweet taste. Future research
Another area of research focuses on improving the heat stability of aspartame. Using encapsulation technology, aspartame has been developed which can be used in baked goods and baking mixes. Initial test results are positive, and FDA approval has been granted for bakery applications.
Since only three synthetic sugar substitutes are currently approved for use in food in the U.S., combining artificial sweeteners in products is becoming an important technological advance. Here, scientists combine two or three sweeteners in an effort to make the product taste more sugarlike.
Where to Learn More
Books
Nabors, Lyn, and Robert Gelardi. Alternative Sweeteners. Marcel Dekker, Inc., 1986.
Periodicals
Best, Daniel and Lisa Nelson. "Low-calorie foods and sweeteners." Prepared Foods, June 1993, p. 47.
Tomasula, Dean. "Sweet as sugar: artificial sweetener producers are blending products, in search of a market winning combination." Chemical Marketing Reporter, June 27, 1994, p. S22.
— Perry Romanowski