Syringe
The hypodermic syringe, also known as the hypodermic needle, is a device used by medical professionals to transfer liquids into or out of the body. It is made up of a hollow needle, which is attached to a tube and a plunger. When the plunger handle is pulled back, fluids are drawn into the tube. The fluid is forced out through the needle when the handle is pushed down. The syringe was introduced in the mid 1800s and has steadily improved with the development of new materials and designs. Today, it has become such an important medical tool that it is nearly a symbol synonymous with the practicing physician.
History
Since the advent of pharmaceutical drugs, methods for administering those drugs have been sought. Various important developments needed to occur before injections through a hypodermic syringe could be conceived. Early nineteenth century physicians were not aware that drugs could be introduced into the body through the skin. One early experiment that demonstrated this idea, however, was performed by Francois Magendie in 1809. In his published work, he outlined a method for introducing strychnine into a dog by using a coated wooden barb. In 1825, A. J. Lesieur described another method for administering drugs through the skin, applying them directly to blisters on the skin. Expanding on results from these experiments, G. V. Lafargue developed a procedure for introducing morphine under the skin using a lancet. A drip needle was invented by F. Rynd in 1844 for the same purpose. However, he did not publish his method until 1861, eight years after the first hypodermic syringe was described.
The first true hypodermic syringe was created by Alexander Wood in 1853. He modified a regular syringe, which at that time was used for treating birthmarks, by adding a needle. He then used this new device for introducing morphine into the skin of patients who suffered from sleeping disorders. A few years later, he added a graduated scale on the barrel and a finer needle. These modifications were enough to attract the attention of the rest of the medical community, resulting in its more widespread use.
Over the years hypodermic syringes have undergone significant changes that have made them more efficient, more useful, and safer. One such improvement was the incorporation of a glass piston within the cylinder. This innovation prevented leaks and reduced the chances of infections, making the device more reliable. The technology for the mass production of hypodermic syringes was developed in the late nineteenth century. As plastics developed, they were incorporated into the design, reducing cost and further improving safety.
Background
The way in which a hypodermic needle works is simple. Fluid, such as a drug or blood, is drawn up through a hollow needle into the main tube when the plunger handle is pulled back. As long as the needle tip remains in the fluid while the plunger handle is pulled, air will not enter. The user can determine exactly how much material is in the tube by reading the measuring marks on the side of the tube. The liquid is dispensed out through the needle when the plunger handle is pushed back down.
The term hypodermic syringe comes from the Greek words hypo, meaning under, and derma, meaning skin. These terms are appropriate because they describe exactly how the device functions. The needle is used to pierce the top layer of the skin, and the material in the tube is injected in the layer below. In this subcutaneous layer, most injected materials will be readily accepted into the bloodstream and then circulated throughout the body.
A syringe is one of three primary methods for introducing a drug into the body. The others are transepidermal (through the skin) and oral. Using a hypodermic needle as the method of drug administration has some significant advantages over oral ingestion. First, the drugs are protected from the digestive system. This prevents them from being chemically altered or broken down before they can be effective. Second, since the active compounds are quickly absorbed into the bloodstream, they begin working faster. Finally, it is more difficult for the body to reject drugs that are administered by syringe. Transepidermal drug administration is a relatively new technology, and its effects are generally not as immediate as direct injection.
Design
There are many hypodermic syringe designs available. However, all of them have the same general features, including a barrel, plunger, needle, and cap. The barrel is the part of the hypodermic needle that contains the material that is injected or withdrawn. A movable plunger is contained within this tube. The width of the barrel is variable. Some manufacturers make short, wide tubes, and others make long, thin ones. The exact design will depend to some extent on how the device will be used. The end of the barrel to which the needle is attached is tapered. This ensures that only the desired amount of material will be dispensed through the needle. At the base of the barrel away from the needle attachment, two arms flare out. These pieces allow the needle user to press on the plunger with the thumb while holding the tube in place with two fingers. The other end of the barrel is tapered.
The plunger, which is responsible for creating the vacuum to draw up materials and then discharge them, is made of a long, straight piece with a handle at one end and a rubber plunger head on the other. The rubber head fits snugly against the walls of the barrel, making an airtight seal. In addition to ensuring an accurate amount of material is drawn in, the squeegee action of the plunger head keeps materials off the inner walls of the tube.
The needle is the part of the device that actually pierces the layers of the skin. Depending on how deep the injection or fluid extraction will be, the needle orifice can be thinner or wider, and its length varies. It can also be permanently affixed to the body of the syringe or interchangeable. For the latter type of system, a variety of needles would be available to use for different applications. To prevent accidental needle stick injuries, a protective cap is placed over the top of the needle when it is not in use.
Raw Materials
Since hypodermic syringes come in direct contact with the interior of the body, government regulations require that they be made from biocompatible materials which are pharmacologically inert. Additionally, they must be sterilizable and nontoxic. Many different types of materials are used to construct the wide variety of hypodermic needles available. The needles are generally made of a heat-treatable stainless steel or carbon steel. To prevent corrosion, many are nickel plated. Depending on the style of device used, the main body of the tube can be made of plastic, glass, or both. Plastics are also used to make the plunger handle and flexible synthetic rubber for the plunger head.
The Manufacturing
Process
There are many manufacturers of hypodermic needles, and while each one uses a slightly different process for production, the basic steps remain the same, including needle formation, plastic component molding, piece assembly, packaging, labeling, and shipping.
Making the needle
-
1 The needle is produced from steel, which is first heated until it is
molten and then
drawn through a die designed to meet the size requirements of the needle. As it moves along the production line, the steel is further formed and rolled into a continuous, hollow wire. The wire is appropriately cut to form the needle. Some needles are significantly more complex and are produced directly from a die casting. Other metal components on the needle are also produced in this manner.
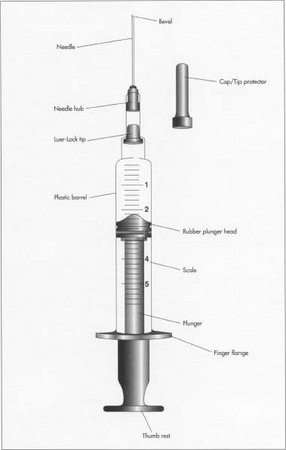 Diagram of a hypodermic syringe. Retraction of the plunger creates the vacuum to draw up materials, which can then be discharged by pushing on the plunger. Its rubber head makes an airtight seal against the walls of the barrel.
Diagram of a hypodermic syringe. Retraction of the plunger creates the vacuum to draw up materials, which can then be discharged by pushing on the plunger. Its rubber head makes an airtight seal against the walls of the barrel.
Making the barrel and plunger
- 2 There are various ways that the syringe tube can be fashioned, depending on the design needed and the raw materials used. One method of production is extrusion molding. The plastic or glass is supplied as granules or powder and is fed into a large hopper. The extrusion process involves a large spiral screw, which forces the material through a heated chamber and makes it a thick, flowing mass. It is then forced through a die, producing a continuous tube that is cooled and cut.
- 3 For pieces that have more complex shapes like the ends, the plunger, or the safety caps, injection molding is used. In this process the plastic is heated, converting it into a liquid. It is then forcibly injected into a mold that is the inverse of the desired shape. After it cools, it solidifies and maintains its shape after the die is opened. Although the head of the plunger is rubber, it can also be manufactured by injection molding. Later, the head of the plunger is attached to the plunger handle.
Assembly and packaging
- 4 When all of the component pieces are available, final assembly can occur. As the tubes travel down a conveyor, the plunger is inserted and held into place. The ends that cap the tube are affixed. Graduation markings may also be printed on the main tube body at this point in the manufacturing process. The machines that print these markings are specially calibrated to ensure they print measurements on accurately. Depending on the design, the needle can also be attached at this time, along with the safety cap.
- 5 After all of the components are in place and printing is complete, the hypodermic syringes are put into appropriate packaging. Since sterility of the device is imperative, steps are taken to ensure they are free from disease-causing agents. They are typically packaged individually in airtight plastic. Groups of syringes are packed into boxes, stacked on pallets, and shipped to distributors.
Quality Control
The quality of the components of these devices are checked during each phase of manufacture. Since thousands of parts are made daily, complete inspection is impossible. Consequently, line inspectors randomly check components at fixed time intervals to ensure they meet size, shape, and consistency specifications. These random samples give a good indication of the quality of the hypodermic syringe produced. Visual inspection is the primary test method. However, more rigorous measurements are also performed. Measuring equipment is used to check the length, width, and thickness of the component pieces. Typically, devices such as a vernier caliper, a micrometer, or a microscope are used. Each of these differ in accuracy and application. In addition to specific tests, line inspectors are stationed at various points of the production process and visually inspect the components as they are made. They check for things such as deformed needles or tubes, pieces that fit together incorrectly, or inappropriate packaging.
Hypodermic syringe production is strictly controlled by the United States government, specifically the Food and Drug Administration (FDA). They have compiled a list of specifications to which every manufacturer must comply. They perform inspections of each of these companies to ensure that they are following good manufacturing practices, handling complaints appropriately, and keeping adequate records related to design and production. Additionally, individual manufacturers have their own product requirements.
The Future
Since Alexander Wood introduced the first device, hypodermic syringe technology has greatly improved. Future research will focus on designing better devices that will be safer, more durable, more reliable, and less expensive to produce. Also, improvements in device manufacture will also continue. One example of this is the trend toward utilizing materials such as metals and plastics that have undergone a minimum of processing from their normal state. This should minimize waste, increase production speed, and reduce costs.
Where to Learn More
Books
Chicka, C. and Anthony Chimpa. Diabetic's Jet Ejectors. Diabetic Gun for Personal Insulin Injection. H.W. Parker, 1989.
Trissel, Lawrence. Pocket Guide to Injectable Drugs: Companion to Handbook of Injectable Drugs. American Society of Health-System Pharmacists, 1994.
— Perry Romanowski
i want to know
how and how much
thank u
CS