Breath Alcohol Tester
Background
There is a serious need to ensure that alcohol-impaired drivers stay off the roads. It is estimated that one person is killed every 32 minutes and another person is injured every 26 seconds in alcohol-related accidents. Highway deaths increased slightly from 41,717 deaths in 1999 to 41,812 in 2000. Forty percent (16,725) involved alcohol, an increase from 38% the previous year.
Breath alcohol testers (BATs) depend on the blood to breath ratio. This ratio describes the relationship between the alcohol content of the breath and the alcohol content of blood at any given time. The accepted ratio of breath alcohol to blood alcohol is 2,100:1. This means 2,100 ml of a deep air lung sample contains the same amount of alcohol as I ml of blood. All breath testing instruments developed since 1939 use this ratio. The ratio is guided by Henry's Law, which states the quantity of gas that dissolves in a liquid at a standard temperature and pressure are directly proportional to the partial pressure of that gas in the gas phase.
In order to obtain an accurate reading of a person's alcohol content the device must test a person's deep lung air. The exchange of gasses, such as alcohol, between the blood and lung occurs in the alveoli. Each lung contains several million alveoli. If deep lung air is not exhaled into the device, the sample breath could be diluted with a lower alcohol concentration. Instruments usually require a person to blow for a minimum amount of time to ensure the air is captured from the deep lungs.
There are two main types of BATs used today. The first type uses infrared light to detect alcohol content. This device passes a breath sample through a narrow band of infrared light set to a frequency absorbed by alcohol. The amount of infrared light not absorbed by the alcohol tells the concentration of alcohol in the breath.
The second type of device uses fuel cells (which rely on chemical reactions) and is the most commonly used BAT. The alcohol in a person's breath is the energy for the fuel cell. The higher the concentration of breath alcohol, the more electricity that will be generated. The device measures the strength of the current to determine the breath/blood alcohol content (BAC).
The fuel cell itself varies only slightly from product to product and is a component that manufacturers purchase from outside venders. In a fuel cell, two electrodes are immersed in a liquid electrolyte, a substance that conducts electricity. An electrode is a solid electric conductor through which an electric current enters or leaves. The electrodes are coated with a platinum layer and have very fine pores. Between the electrodes is the thin electrolyte layer. The alcohol is pulled into the fuel cell by a pump and seeps through the electrodes, where it is then chemically converted. The fuel results in a flow of electricity between the electrodes.
The alcohol found in alcoholic beverages is ethyl alcohol, also known as ethanol. The molecular structure of ethanol has four major types of bonds: carbon to oxygen, carbon to carbon, carbon to hydrogen, and oxygen to hydrogen. In a fuel cell tester, the platinum material on the electrodes acts as a chemical catalyst and ionizes the hydrogen atoms by taking away their electrons. The hydrogen atoms are now positive. In this chemical conversion (which takes place at the top of the fuel cell) the hydrogen atoms then move lower in the fuel cell and combine with oxygen. Water is formed and one electron per positive hydrogen molecule is absorbed. Now there are more electrons at the top of the cell than at the bottom. The two surfaces are connected electrically through a wire. The electrons flow through the wire from the platinum electrode. The wire is connected to an electrical-current meter and to the platinum electrode on the other side. The result is a neutralizing current that flows through the fuel cell. The current indicates the amount of alcohol consumed by the fuel cell. The more fuel (alcohol) present, the higher the current.
History
Since the time of Hippocrates (c. 430 B.C. ), physicians have known that human breath can provide clues to a medical diagnosis. Breath is one accurate way to measure a person's BAC because blood goes through the lungs, the site of gas exchange. The alcohol molecules are transferred from the blood to the lung air expelled in a breath.
In 1938, the first BAT was developed by Dr. R. N. Harger and called the Drunkometer. The year 1941 brought about the Intoximeter invented by Glenn Forrester and then the Alcometer developed by Professor Leon Greenberg. These machines calculated the blood alcohol to breath alcohol levels of deep lung air samples. The only way to determine the BAC before these instruments was through blood or urine tests. These methods were both time consuming and expensive. In 1954, the Breathalyzer was invented by Indiana State Policeman Robert Borkenstein. This was a portable, durable type of alcohol testing device that became the instrument of choice by police around the country.
Early breath alcohol testers required the person being tested to blow up a balloon. This ensured that a deep air lung sample would be taken. The balloon air was then released over photoelectric chemicals that changed color in the presence of alcohol; the deeper the color change, the higher the alcohol content. This device was often challenged in court because it could produce false results. For example, if a person used a mouthwash containing alcohol before taking the test, it could result in a higher BAC.
By the 1980s chemical breath tests were rarely used. Suspects were continuously challenging the results and courts were overturning them. Manufacturers focused on improving the accuracy, speed, and ease of fuel and infrared BAT use.
Raw Materials
Fuel cell breath alcohol testers are primarily composed of a fuel cell, pump, mouthpiece, printed circuit board (PCB), and a liquid crystal display (LCD) or light-emitting diode (LED) all contained within a plastic case generally made from low density polyethylene (LDPE), polypropylene (PP), or polystyrene (PS) plastic. The fuel cell is made from two platinum coated electrodes and a permeable electrolyte material. The pump is made of glass and nylon and used to pull the alcohol into the BAT. At the heart of the BAT is the PCB which controls the entire unit. The microprocessor contains the coding that the BAT uses to carry out the functions. The device also uses a LCD or LED to present instructions to the user, including the results or potential error messages.
Design
Both infrared and fuel-cell breath testing devices are used in three different types of instruments: evidential, screening, and passive breath alcohol testers. Evidential BATs collect samples from a person's breath. The results are accurate enough that the collected evidence can be used in a court of law. For the most part, these devices are large—the size of a desktop computer—and housed at the police department. Screening BATs are typically the most widely used BAC testers due to their accuracy and portability (they are about the size of a pocket calculator). These BAC testers require a person to blow into the device, and it reads either pass, fail, or provides a digital readout of the person's BAC. Passive BATs are also handheld devices, but require no action on the suspect's part. The device takes samples of the air around a person.
The Manufacturing
Process
-
The first step in the production of a fuel cell BAT is to manufacture
the case.
Plastic—LDPE, PP, or PS—pellets are fed into a hopper and heated until they are fully liquidized.
 A fuel cell breath alcohol tester.
A fuel cell breath alcohol tester. - The hopper then releases the viscous liquid into a plastic injection mold. In this process, the plastic is poured into a die that is shaped like the desired case. After the liquid fills the die, it is closed and any excess material is drained. After the die cools, it opens and the case is ejected.
- The PCB is made from a fiberglass epoxy resin with a copper coil bonded to one or both sides. These arrive at the plant assembled.
- Two springs are soldered to the PCB that will connect to the batteries. When the case is closed, the battery compartment on the case lines up with the springs so the batteries are ready to fit in the compartment.
- The pump is what draws a person's breath inside the testing device to the fuel cell. The manufacturer takes a three-volt motor, Pyrex cylinder, nylon piston, and a stainless steel lead screw. The pump turns the stainless steel screw and the screw moves the pistons back and forth, forcing the breath through the fuel cell. Pyrex is a heat-resistant, chemical-resistant glass. The nylon piston is a solid cylinder or disk that fits snugly into the cylinder. An o-ring, a small ring made of rubber or plastic, is manually placed on the groove of the piston. The piston is then lubricated slightly then fit inside the Pyrex cylinder. Workers make sure the pieces are firmly held together. They then attach the motor and the lead screw to the pump with bolts.
- After the pump is assembled it is tested on a test jig, a contraption that manufacturers build to test products. Every manufacturer has their own unique test jig but the goal is the same for all: to ensure the pump can properly cycle air in and out numerous times. Workers hook the pump to a machine that releases air through the pump about 200 times. They monitor the air current passing through the pump and make sure it is within specifications. Specifications require a set quantity of air moving through the device.
- Once the pump is complete the rest of the pieces are assembled into the plastic case. The PCB is manually screwed into the case. Then the LCD is connected to the PCB. The LCD has a connector on one side. The connector is attached to a flexible strip consisting of wires and a flat piece of tape-like material. Workers push the connector into the PCB and it snaps into place.
- Next, workers fasten the pre-assembled fuel cell to the rest of the BAT components. The fuel cell is attached to a porting block. The porting block is the plumbing of the device (a small plastic tube that the sample air moves in as it heads towards the fuel cell). Workers attach the fuel cell to the porting block with four screws. The porting block is then fixed to the microprocessor with two screws. The pump is attached to the porting block with piece of silicon tubing. The tubing ties the pump to the porting assembly.
-
Once the BAT is completely assembled manufacturers need to input
measurements into the device. Manufacturers set the device by a process
called calibration. Calibration is the process of setting-up the BAT to
a known, standard alcohol level in order to accurately measure the
alcohol concentration in the breath. The BAT will sample either a test
gas or solution of a known alcohol concentration, called the alcohol
standard. Once tested, the BAT will automatically
adjust all internal parameters to provide accurate measurement.
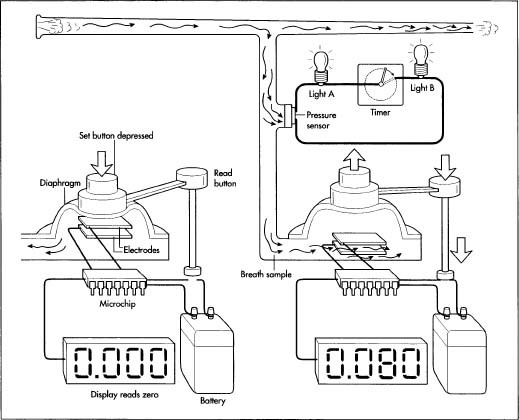 An example of how a fuel cell breath alcohol tester works.
An example of how a fuel cell breath alcohol tester works. - After the BATs have been tested, they are packaged and shipped to the wholesaler or directly to individual customers.
Quality Control
Each BAT has a self-diagnosis test built into the PCB. Manufacturers test all the functions of the device. The units are cycled through simulators and tested repeatedly. Manufacturers can run a cycle of 50 tests at once. For example, the PCB is programmed to beep when the device has detected enough deep lung air entering it. It is also programmed to graph the air being blown.
Manufacturers will push a known amount of air through the machine to test multiple functions at once.
When manufacturers have completed their testing, they send their product to the United States Department of Transportation. The Department of Transportation must approve a device before placing it on their Conforming Products list. In terms of law enforcement, states are required to purchase only devices on the Conforming Products list. Government agencies will also only purchase devices from those approved by the Department. As these markets are the vast majority of alcohol testing devices sold in the United States, manufacturers regularly send their product to a Department of Transportation office for approval. The Department of Transportation tests the device for performance. Laboratory workers use a wet or gas solution with a known concentration of alcohol. If using a wet bath, the device is hooked up to an instrument called a wet bath simulator. The solution is poured into the simulator and the instrument automatically heats the solution to a temperature of 93°F (34°C). The simulator has a gas outlet port. Workers connect a mouthpiece adapter to the port, then connect Bat's mouthpiece to the mouthpiece adapter. Vapor is pushed from the wet sample simulator into the BAT at a constant rate throughout the cycle. Workers then program the unit to 0.01. Testers can determine the exact concentration of alcohol that emerges from the solution by knowing the temperature. The simulator pushes vapor through the device and testers match up what the device reads with the known alcoholic concentration. BATs are required to meet specifications that are set by the Department. For example, among the requirements made by the Department of Transportation is that the device must distinguish alcohol from acetone when alcohol is at a 0.02 level. Laboratory workers repeat the test roughly 200 times, using different test conditions. They vary the known temperature from cold to warm, such as 50'F, 68°F, and 86°F (10°C, 20°C, and 30°C).
The Department of Transportation also ensures that enforcement personnel accurately use the alcohol-testing device. Law enforcement personnel must pass a certification that conduct breath alcohol testing by either their state or local government.
The Future
The push is to manufacture portable devices that can collect evidence on the spot. To accomplish this, there is an increasing move towards manufacturing alcohol fuel cell devices. As opposed to alcohol infrared devices, the fuel cells testers are much less expensive. Their accuracy and ease of use is comparable to infrared testers.
The desire for hand-held BATs made for the individual consumer market is also growing. People who want to know if they are over the legal alcohol limit can test themselves with these BATs. Establishments that sell alcohol also are a potential market for these devices. These products are about the size of a flashlight and far less expensive than the devices geared for professionals. They are not as precise as other testing devices, but have the benefit of ease and cost.
Law enforcement personnel are one of the main groups who commonly and regularly use breath testing devices to ensure drivers are not under the influence of alcohol. These devices are also used to ensure alcohol abuse does not occur in the workplace. The United States Department of Transportation requires alcohol testing for millions of safety-sensitive employees in the trucking, airline, rail, transit, and maritime industries. Testing of these employees is administered randomly. Non-regulated companies are increasingly administering alcohol tests on their employees.
In 2001, most states have a legal limit of a 0.1 BAC, with some states enforcing a 0.08. That translates to the BAC not exceeding 10 or 8 mg per 100 ml of blood.
Where to Learn More
Books
Considine, Douglas M., ed. Van Nostrand's Scientific Encyclopedia. 8th ed. New York: International Thomson Publishing Inc., 1995.
Periodicals
National Center for Statistics and Analysis. Motor Vehicle Traffic Crash Fatality and Injury Estimates for 2000, 2000 Annual Assessment of Motor Vehicle Crashes.
Other
"About Breath Tests." Menssanna Research, Inc. Web Page. December 2001. < http://www.menssanaresearch.com >.
Craig C. Freudenrich. "How Breathalyzers Work." How Stuff Works Web Page. December 2001. < http://www.howstuffworks.com/breathalyzer >.
Life Loc Technologies, Inc. Web Page. December 2001. < http://www.lifeloc.com >.
M. Rae Nelson