Hairsproy
Background
Hairsprays belong to a class of personal care products that help hair to hold a desired style. These products contain film forming ingredients that are applied as a fine mist. When dry, these chemicals form tiny glue-like spots that hold the hair shafts together. Hairsprays are formulated as aerosols that are powered by pressurized gasses or non-aerosols that are dispensed by manually depressing a pump.
History
Women have used natural compounds (such as clays and gums) to hold their hair in place since antiquity. The modern hairspray was not born until aerosol spray containers were developed by the United States Army during World War II. The army created this technology to enable them to spray insecticides over large areas. Over the next decade or so, aerosol technology spread to other industries such as paints and coatings and personal care. The first commercial hairsprays were marketed in the late 1940s. These early products used shellac, a natural resin, to hold hair in place. In the last 50 years researchers have improved both the quality of hairspray ingredients and the aerosol packaging used to deliver them. Hairsprays have since become an important part of the hair care market.
Raw Materials
Holding agents
Hairsprays work by coating the hair with polymers, which are long chain chemical compounds. Polymers are sometimes called resins because natural resin materials (like shellac) were used in the first hair sprays. The key property that makes polymers useful as hair holding agents is their ability to form films upon drying. Once hair spray is applied to the hair, the liquid drops run down the hair shaft until they reach the intersection of two hair fibers. When the drops dry at this fiber intersection, they create an invisible film that bonds hairs together.
Chemists may choose from many different polymers when developing hair styling products. For example, polyvinylpyrrolidone (also known as PVP) is a common polymer used in hair styling products. However, it does not provide a strong hold and it tends to pick up moisture from the air. To remedy this situation chemists combine PVP with another polymer, vinyl acetate. The resulting mixture, PVPVA, is a copolymer that has improved humidity resistance and will therefore hold curls better. However, if not properly formulated PVPVA copolymers can be so waterproof that they become difficult to wash out of the hair. Another common copolymer is made from vinyl acetate and crotonic acid. This ingredient is popular because it provides the proper balance of hardness, solubility, and moisture susceptibility.
Solvents
Solvents make up the largest portion by weight of an aerosol hairspray. They are used as a carrier for the active ingredients in the formulation and are selected based on their compatibility with the chemical actives and the propellant. Water is a popular solvent due to its low cost. Unfortunately, formulations that contain water take longer to dry and are less soluble in many propellant systems. Water also increases the chance of corrosion inside the can. Ethanol, although somewhat more expensive, is another popular solvent. However, ethanol belongs to the class of volatile organic compounds (VOCs) whose use in aerosols have been restricted because they contribute to air pollution. To date, no acceptable replacement solvents have been approved.
Additives
Hairsprays contain a number of chemical additives in addition to polymers and solvents. For example, plasticizers are added to modify the effects of polymers. These include chemicals such as isopropyl myristate, diethyl phthalate, and silicones that can make hairspray films more flexible and less brittle. Neutralizing and anti-corrosion agents, like aminomethyl propanol (AMP), ammonium hydroxide, morpholine, cyclohexylamine, and borate esters are added to control resin solubility and help prevent the inside of the can from rusting.
Propellants
Propellants, as the name implies, are responsible for propelling the hairspray out of the can. These are gasses that can be stored under low pressure in the can. Originally chlorofluorocarbon gasses (CFCs) were used, but they have been banned due to their suspected complicity in the depletion of the ozone layer in the upper atmosphere. Hydrocarbon propellants like butane and propane were used as replacements for CFCs. These gasses are mixtures of butane and propane designed to deliver a certain amount of pressure in the can. For example, the propellant Butane 40 is a mixture of butane and propane that has a vapor pressure of 40 lb/in 2 (18 kg/cm 2 ). Hydrocarbon propellants were extremely popular until the 1980s when California and a few other American states began to legislate how much of these gasses could be used in hairsprays because they were shown to contribute to air pollution. This legislation led to decreased use of hydrocarbons.
Scientists working at Dupont developed a new class of propellants. These are known as hydrofluorocarbons (HFCs), and they have many of the properties of CFCs but are not as polluting. Popular HFCs are 1,1,-difluoroethane (Propellant 152A) and 1, 1, 1, 2,-tetrafluoromethane (Propellant 134A). While they are relatively expensive, they are used to formulate fast drying hairsprays.
Packaging
Aerosol hairsprays have traditionally been packaged in containers made from tin plated steel or aluminum. The package is fitted with a valve that both seals the can and dispenses the contents. The valve is connected to a plastic dip tube that carries the liquid product up through the can to the top. The upper portion of the valve is fitted with a button, or actuator, that is pressed to open the valve and release the product.
The valve itself is a very complicated piece of equipment. It consists of a body section with a tail piece that attaches to the diptube. The central part of the valve body serves as a mixing chamber where the propellant and liquid concentrate blend together. This mixing process is very turbulent and helps break the hairspray into very fine mist. At the top of the valve body is a stem that feeds product from the mixing chamber into the dispensing button. The entire valve assembly is housed in a metallic cup which is a ring shaped piece of either aluminum or tinplate steel.
Design
Chemists must consider several key parameters when designing hairspray formulations including efficacy, safety, cost, consumer appeal, and regulatory considerations. The functionality of the product is a key consideration. Formulating chemists have hundreds, if not thousands, of ingredients to choose from. They begin by selecting a resin that will give the desired hold characteristics and combine it with plasticizers and other ingredients that give the proper feel on the hair.
For a polymer to make a good styling resin, it must have certain characteristics. It must deposit a film that is substantive on the hair yet that can still be easily washed away; it must hold hair with flexibility so the hair can move without breaking the film; it must be transparent so it does not reduce the hair's natural gloss; it should not flake when the hair is brushed; and it must not absorb moisture from the atmosphere and become sticky.
Because the ingredients used in hairsprays may be in contact with the skin for an extended period of time, they must be designed to be non-irritating and non-sensitizing. To make sure they are not hazardous when in-haled,
Chemists design hairsprays to be more consumer appealing by employing pleasant fragrances and easy to use packaging. Some fragrances are designed to simply cover the odor of the chemicals in the formulation; others are designed to be highly fragrant. Packaging design options also help increase the product's aesthetic appeal. These include easy to use dispensers like toggle valves and large actuator buttons.
Typically hairsprays are designed to deliver 8 oz (237 ml) of product but both smaller and larger sizes are available. The pressure in the can is generally between 40 and 80 lb/in 2 (18 and 36 kg/cm 2 ) depending on the type of propellant that is used. It is also important to note that hairsprays are designed to have a minimum shelf life of three years but have been known to last five years and longer.
The Manufacturing
Process
The manufacture of aerosol hairspray requires highly automated equipment. Because of the cost involved operating this equipment, most hairspray marketers choose to have their products produced by specialized vendors known as contract manufacturers.
Batching
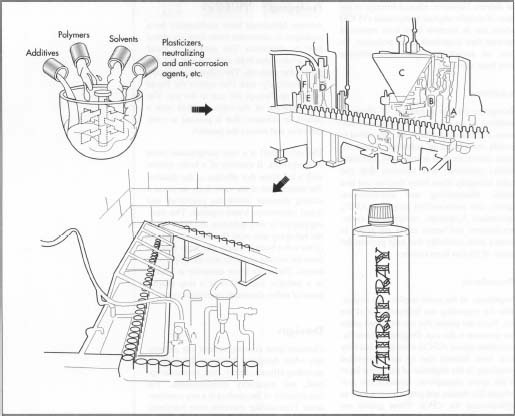
- 1 During the manufacturing process the formula is prepared as a liquid concentrate in large batch tanks composed of stainless steel, coated aluminum, or fiber glass reinforced polyester. These batches may be as large as 2,000 gal (7,5701). The tanks are equipped with a large turbine mixer with blades that are several feet in diameter. The solvent is charged into the tank first and then followed by the other ingredients. The solvent makes up the largest proportion of the formula and may be present at 80% by weight or greater. Other ingredients range in concentration from a few tenths of a percent for some of the pH control agents, to a few percent for fragrance, to approximately 10% for the resin. Depending on the solubility of the ingredients in given formula, this mixing step may take as little as 30 minutes or as long as several hours. Since some of the ingredients are in the form of powders, the mixture must be carefully monitored during the batching process to ensure they dissolve properly. After mixing is complete the concentrate is tested to ensure it conforms to specifications and is then transferred to a holding tank prior to filling.
Filling
- 2 The packaging components are staged on the filling machinery. As the empty cans move down the conveyor belt, a jet of compressed air removes any dirt and dust.
- 3 At the next stage of the filling line, there are a series of nozzles, known as filling heads, that are connected to tubes that transfer the liquid concentrate from the tank where it is stored. A piston mechanism injects a precise amount of liquid into the can.
- 4 The cans proceed down the line to the next station where two actions occur at once. The gaseous propellant is shot into the cans and the valve cup is immediately crimped into place. The metal cup is crimped onto the rim on the opening of the can. This tight seal prevents the gas and liquid from leaking out.
- 5 After the gassing operation the cans are fed through a long trough filled with hot water. As the cans slowly move underwater they are checked visually for escaping bubbles, which would indicate a bad valve seal or a leaky can. Leaking cans are removed during this stage of the operation.
- 6 After exiting the water-bath, cans are dried by compressed air jets. A cap is placed over the valve at the end of the filling line; this prevents the aerosol from being accidentally activated during shipping.
- 7 Finally the finished units are packed into boxes and stacked on pallets for shipping.
Byproducts/Waste
The most obvious byproducts of aerosol hairspray are the environmentally unfriendly VOCs emitted into the atmosphere. In addition to depleting the ozone layer, these chemical byproducts are also used in inhalant abuse. Although companies have developed alternatives (such as manual pump sprays), many consumers prefer and continue to use the aerosol can.
Quality Control
During the manufacturing process, samples are pulled from the conveyor line and checked to ensure they meet all specifications. Key properties that are monitored include the fill weight, the concentration of active ingredients, and the pressure of the can. Spray characteristics such as spray rate (the amount of product delivered per second) and the spray pattern (the physical size and shape of the spray) are also monitored. Long-term stability testing is conducted to ensure that the cans do not clog when sprayed and that they remain free of internal corrosion.
The Future
The future of hairsprays depends not only market considerations but also on the regulatory actions of state and federal agencies. Since the 1970s, the industry has struggled with the severe limitations imposed by the government. To circumvent these limitations, researchers continue to experiment with polymers that can be incorporated into non-polluting, water-based formulas. In addition, they are evaluating several non-traditional aerosol delivery systems that release fewer contaminants into the atmosphere. Pump sprays are the most well-known alternative to aerosols, and they operate by the physical force generated when a spring inside the pump is compressed. Other aerosol alternatives include the bag-in-the-can system that uses the physical force of a stretched rubber bladder to spray the contents. Hairspray marketers will have to continue to find cutting edge technology like this to keep pace with an ever changing regulatory environment.
Where to Learn More
Books
Dallal, Joseph, and Colleen Rocafort. Hair Styling/Fixative Products, in Hair and Hair Care. Marcel Dekker, 1997.
Schueller, Randy, and Perry Romanowski. Beginning Cosmetic Chemistry. Allured Publishing, 1999.
Randy Schueller