Soft Drink
Background
Soft drinks are enormously popular beverages consisting primarily of carbonated water, sugar, and flavorings. Nearly 200 nations enjoy the sweet, sparkling soda with an annual consumption of more than 34 billion gallons. Soft drinks rank as America's favorite beverage segment, representing 25% of the total beverage market. In the early 1990s per capita consumption of soft drinks in the U.S. was 49 gallons, 15 gallons more than the next most popular beverage, water.
The roots of soft drinks extend to ancient times. Two thousand years ago Greeks and Romans recognized the medicinal value of mineral water and bathed in it for relaxation, a practice that continues to the present. In the late 1700s Europeans and Americans began drinking the sparkling mineral water for its reputed therapeutic benefits. The first imitation mineral water in the U.S. was patented in 1809. It was called "soda water" and consisted of water and sodium bicarbonate mixed with acid to add effervescence. Pharmacists in America and Europe experimented with myriad ingredients in the hope of finding new remedies for various ailments. Already the flavored soda waters were hailed as brain tonics for curing headaches, hangovers, and nervous afflictions.
Pharmacies equipped with "soda fountains" featuring the medicinal soda water soon developed into regular meeting places for local populations. Flavored soda water gained popularity not only for medicinal benefits but for the refreshing taste as well. The market expanded in the 1830s when soda water was first sold in glass bottles. Filling and capping the gaseous liquid in containers was a difficult process until 1850, when a manual filling and corking machine was successfully designed. The term "soda pop" originated in the 1860s from the popping sound of escaping gas as a soda bottle was opened.
New soda flavors constantly appeared on the market. Some of the more popular flavors were ginger ale, sarsaparilla, root beer, lemon, and other fruit flavors. In the early 1880s pharmacists experimented with powerful stimulants to add to soda water, including cola nuts and coca leaves. They were inspired by Bolivian Indian workers who chewed coca leaves to ward off fatigue and by West African workers who chewed cola nuts as a stimulant. In 1886 an Atlanta pharmacist, John Pemberton, took the fateful step of combining coca with cola, thus creating what would become the world's most famous drink, "Coca-Cola". The beverage was advertised as refreshing as well as therapeutic: "French Wine Cola—Ideal Nerve and Tonic Stimulant." A few years later another pharmacist, Caleb Bradham, created "Pepsi-Cola" in North Carolina. Although the name was a derivation of pepsin, an acid that aids digestion, Pepsi did not advertise the beverage as having therapeutic benefits. By the early 20th century, most cola companies focused their advertising on the refreshing aspects of their drinks.
As flavored carbonated beverages gained popularity, manufacturers struggled to find an appropriate name for the drinks. Some suggested "marble water," "syrup water," and "aerated water." The most appealing name, however, was "soft drink," adapted in the hopes that soft drinks would ultimately supplant the "hard liquor" market. Although the idea never stuck, the term soft drink did.
Until the 1890s soft drinks were produced manually, from blowing bottles individually to filling and packaging. During the following two decades automated machinery greatly increased the productivity of soft drink plants. Probably the most important development in bottling technology occurred with the invention of the "crown cap" in 1892, which successfully contained the carbon dioxide gas in glass bottles. The crown cap design endured for 70 years.
The advent of motor vehicles spawned further growth in the soft drink industry. Vending machines, serving soft drinks in cups, became regular fixtures at service stations across the country. In the late 1950s aluminum beverage cans were introduced, equipped with convenient pull-ring tabs and later with stay-on tabs. Light-weight and break-resistant plastic bottles came into use in the 1970s, though it was not until 1991 that the soft drink industry used plastic PET (polyethylene terephthalate) on a wide scale.
Soft drink manufacturers have been quick to respond to consumer preferences. In 1962 diet colas were introduced in response to the fashion of thinness for women. In the 1980s the growing health consciousness of the country led to the creation of caffeine-free and low-sodium soft drinks. The 1990s ushered in clear colas that were colorless, caffeine-free, and preservative-free.
Raw Materials
Carbonated water constitutes up to 94% of a soft drink. Carbon dioxide adds that special sparkle and bite to the beverage and also acts as a mild preservative. Carbon dioxide is an uniquely suitable gas for soft drinks because it is inert, non-toxic, and relatively inexpensive and easy to liquefy.
The second main ingredient is sugar, which makes up 7-12% of a soft drink. Used in either dry or liquid form, sugar adds sweetness and body to the beverage, enhancing the "mouth-feel," an important component for consumer enjoyment of a soft drink. Sugar also balances flavors and acids.
Sugar-free soft drinks stemmed from a sugar scarcity during World War II. Soft drink manufacturers turned to high-intensity sweeteners, mainly saccharin, which was phased out in the 1970s when it was declared a potential carcinogen. Other sugar substitutes were introduced more successfully, notably aspartame, or Nutra-Sweet, which was widely used throughout the 1980s and 1990s for diet soft drinks. Because some high-intensity sweeteners do not provide the desired mouth-feel and aftertaste of sugar, they often are combined with sugar and other sweeteners and flavors to improve the beverage.
The overall flavor of a soft drink depends on an intricate balance of sweetness, tartness, and acidity (pH). Acids add a sharpness to the background taste and enhance the thirst-quenching experience by stimulating saliva flow. The most common acid in soft drinks is citric acid, which has a lemony flavor. Acids also reduce pH levels, mildly preserving the beverage.
Very small quantities of other additives enhance taste, mouth-feel, aroma, and appearance of the beverage. There is an endless range of flavorings; they may be natural, natural identical (chemically synthesized imitations), or artificial (chemically unrelated to natural flavors). Emulsions are added to soft drinks primarily to enhance "eye appeal" by serving as clouding agents. Emulsions are mixtures of liquids that are generally incompatible. They consist of water-based elements, such as gums, pectins, and preservatives; and oil-based liquids, such as flavors, colors, and weighing agents. Saponins enhance the foamy head of certain soft drinks, like cream soda and ginger beer.
To impede the growth of microorganisms and prevent deterioration, preservatives are added to soft drinks. Anti-oxidants, such as BHA and ascorbic acid, maintain color and flavor. Beginning in the 1980s, soft drink manufacturers opted for natural additives in response to increasing health concerns of the public.
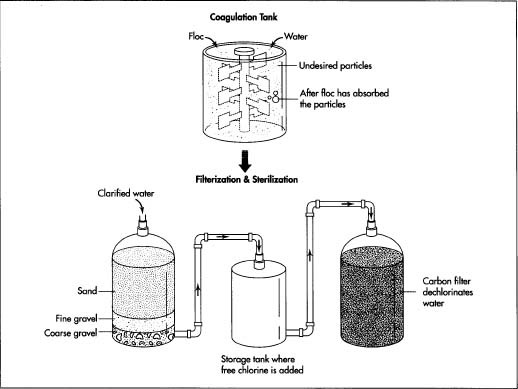
The Manufacturing
Process
Most soft drinks are made at local bottling and canning companies. Brand name franchise companies grant licenses to bottlers to mix the soft drinks in strict accordance to their secret formulas and their required manufacturing procedures.
Clarifying the water
- 1 The quality of water is crucial to the success of a soft drink. Impurities, such as suspended particles, organic matter, and bacteria, may degrade taste and color. They are generally removed through the traditional process of a series of coagulation, filtration, and chlorination. Coagulation involves mixing a gelatinous precipitate, or floc (ferric sulphate or aluminum sulphate), into the water. The floc absorbs suspended particles, making them larger and more easily trapped by filters. During the clarification process, alkalinity must be adjusted with an addition of lime to reach the desired pH level.
Filtering, sterilizing, and dechlorinating the water
- 2 The clarified water is poured through a sand filter to remove fine particles of floc. The water passes through a layer of sand and courser beds of gravel to capture the particles.
-
3 Sterilization is necessary to destroy bacteria and organic compounds
that might spoil the water's taste or color. The water is pumped
into a storage tank and is dosed with a small amount of free chlorine.
The chlorinated water remains in the storage
tank for about two hours until the reaction is complete.
- 4 Next, an activated carbon filter dechlorinates the water and removes residual organic matter, much like the sand filter. A vacuum pump de-aerates the water before it passes into a dosing station.
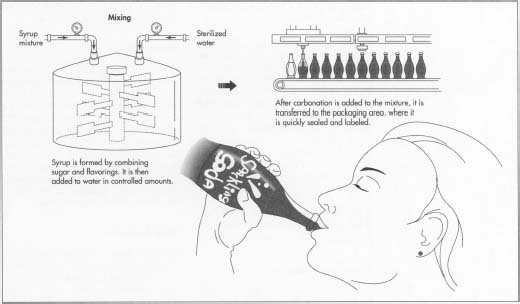
Mixing the ingredients
- 5 The dissolved sugar and flavor concentrates are pumped into the dosing station in a predetermined sequence according to their compatibility. The ingredients are conveyed into batch tanks where they are carefully mixed; too much agitation can cause unwanted aeration. The syrup may be sterilized while in the tanks, using ultraviolet radiation or flash pasteurization, which involves quickly heating and cooling the mixture. Fruit based syrups generally must be pasteurized.
- 6 The water and syrup are carefully combined by sophisticated machines, called proportioners, which regulate the flow rates and ratios of the liquids. The vessels are pressurized with carbon dioxide to prevent aeration of the mixture.
Carbonating the beverage
- 7 Carbonation is generally added to the finished product, though it may be mixed into the water at an earlier stage. The temperature of the liquid must be carefully controlled since carbon dioxide solubility increases as the liquid temperature decreases. Many carbonators are equipped with their own cooling systems. The amount of carbon dioxide pressure used depends on the type of soft drink. For instance, fruit drinks require far less carbonation than mixer drinks, such as tonics, which are meant to be diluted with other liquids. The beverage is slightly over-pressured with carbon dioxide to facilitate the movement into storage tanks and ultimately to the filler machine.
Filling and packaging
- 8 The finished product is transferred into bottles or cans at extremely high flow rates. The containers are immediately sealed with pressure-resistant closures, either tinplate or steel crowns with corrugated edges, twist offs, or pull tabs.
- 9 Because soft drinks are generally cooled during the manufacturing process, they must be brought to room temperature before labeling to prevent condensation from ruining the labels. This is usually achieved by spraying the containers with warm water and drying them. Labels are then affixed to bottles to provide information about the brand, ingredients, shelf life, and safe use of the product. Most labels are made of paper though some are made of a plastic film. Cans are generally pre-printed with product information before the filling stage.
- 10 Finally, containers are packed into cartons or trays which are then shipped in larger pallets or crates to distributors.
Quality Control
Soft drink manufacturers adhere to strict water quality standards for allowable dissolved solids, alkalinity, chlorides, sulfates, iron, and aluminum. Not only is it in the interest of public health, but clean water also facilitates the production process and maintains consistency in flavor, color, and body. Microbiological and other testing occur regularly. The National Soft Drink Association and other agencies set standards for regulating the quality of sugar and other ingredients. If soft drinks are produced with low-quality sugar, particles in the beverage will spoil it, creating floc. To prevent such spoilage, sugar must be carefully handled in dry, sanitized environments.
It is crucial for soft drink manufacturers to inspect raw materials before they are mixed with other ingredients, because preservatives may not kill all bacteria. All tanks, pumps, and containers are thoroughly sterilized and continuously monitored. Cans, made of aluminum alloy or tin-coated low-carbon steel, are lacquered internally to seal the metal and prevent corrosion from contact with the beverage. Soft drink manufacturers also recommend specific storage conditions to retailers to insure that the beverages do not spoil. The shelf life of soft drinks is generally at least one year.
Recycling
The $27 billion dollar soft drink industry generated about 110 billion containers each year in the early 1990s. About half of soft drink containers were aluminum cans and the other half, about 35 billion, were PET plastic bottles. Nearly 60% of all soft drink containers were recycled, the highest rate for any packaging in the United States. Environmental concerns continued to lead to improvements and innovations in packaging technology, including the development of refillable and reusable containers.
The Future
In the 1990s there were more than 450 types of soft drinks on the market and new flavors and sweeteners are developed all the time to meet market demands. In the future, advanced technology will lead to greater efficiency of soft drink production at all stages. New methods of water clarification, sterilization, and pasteurization will improve production and minimize the need for preservatives in soft drinks. Concerns with consumer health, safety, and the environment will continue to have a positive impact on trends in the soft drink industry.
Where To Learn More
Books
Louis, J.C. The Cola Wars. Everest House, 1980.
Mitchell, Alan J., ed. Formulation and Production of Carbonated Soft Drinks. AVI, 1990.
Oliver, Thomas. The Real Coke. Random House, 1986.
Riley, John J. A History of the American Soft Drink Industry. Arno Press, 1972.
— Audra Avizienis
Regards,
Husin
I have this question to ask. Why are soft drinks generally not sterilised? Send the answer to my e-mail
...but i just want to ask if it is true that all soda/soft drinks
have only bad effects in our health??...thanks!
what ACID do you refer?
Good day
my question is :-
what is the best to use in producing soft drink, is it citric acid or malic acid in order to get an extra bite and strong mouth feel bite while drinking a soft drink ?
all what explained above is good , but what is the flavor that shall be added to give a strong bite in mouth and to help co2 to keep strong for long time after open pet bottel ?
dose adding cream soda will give a foum ? & which type of cream soda to add ?
dose cream soda can be easly filled or shall be used anitfoum to fill by filler ?
regards
rami
thanks®ards